chemical polarity
Chemical polarity
Chemical polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end. Polar molecules must contain one or more polar bonds due to a difference in electronegativity between the bonded atoms. Molecules containing polar bonds have no molecular polarity if the bond dipoles cancel each other out by symmetry. Polar molecules interact through dipole-dipole interactions and hydrogen bonds.
It is to note that not all atoms attract electrons with the same force. Atoms with high electronegativity, such as fluorine, oxygen, and nitrogen, exert a greater pull on electrons than atoms with lower electronegativities such as alkali metals and alkaline earth metals. In a bond, this leads to unequal sharing of electrons between the atoms, as electrons will be drawn closer to the atom with the higher electronegativity.
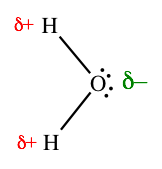
Because electrons have a negative charge, the unequal sharing of electrons within a bond leads to the formation of an electric dipole: a separation of positive and negative electric charge. Because the amount of charge separated in such dipoles is usually smaller than a fundamental charge, they are called partial charges, denoted as \[\delta+\] and \[\delta-\]. The bond dipole moment is calculated by multiplying the amount of charge separated and the distance between the charges. The dipole moment, despite being directly proportion to the distance between charges, is more heavily influenced by the electronegativity difference between the two atoms/ions.
The arrows indicate the direction in which the electron moves (or more accurately, more likely to be found).
Partial charges:
Polar molecules
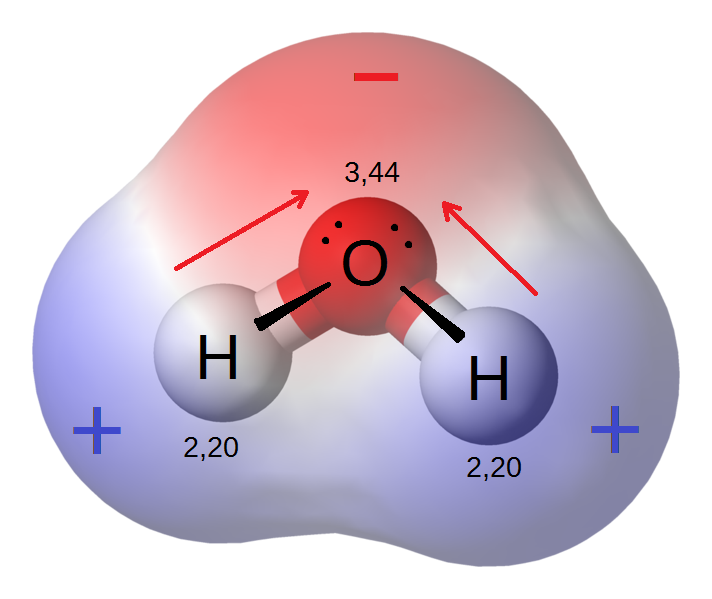
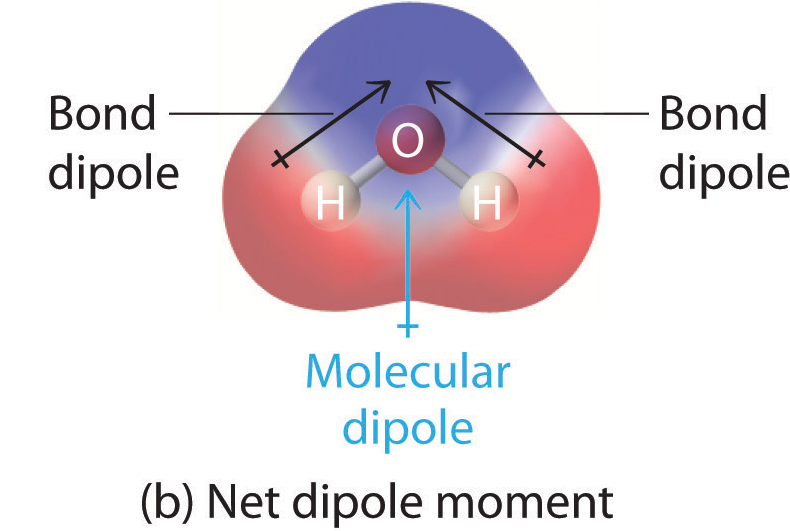
A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (\[\ce{H2O}\]) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. If the bond dipole moments of the molecule do not cancel, the molecule is polar. For example, the water molecule (\[\ce{H2O}\]) contains two polar \[\ce{O-H}\] bonds in a bent (nonlinear) geometry. The bond dipole moments do not cancel, so that the molecule forms a molecular dipole with its negative pole at the oxygen and its positive pole midway between the two hydrogen atoms.
(Blue = partially negative region, Red = partially positive region)
However, molecules such as
Boron trifluoride, \[\ce{BF3}\]: 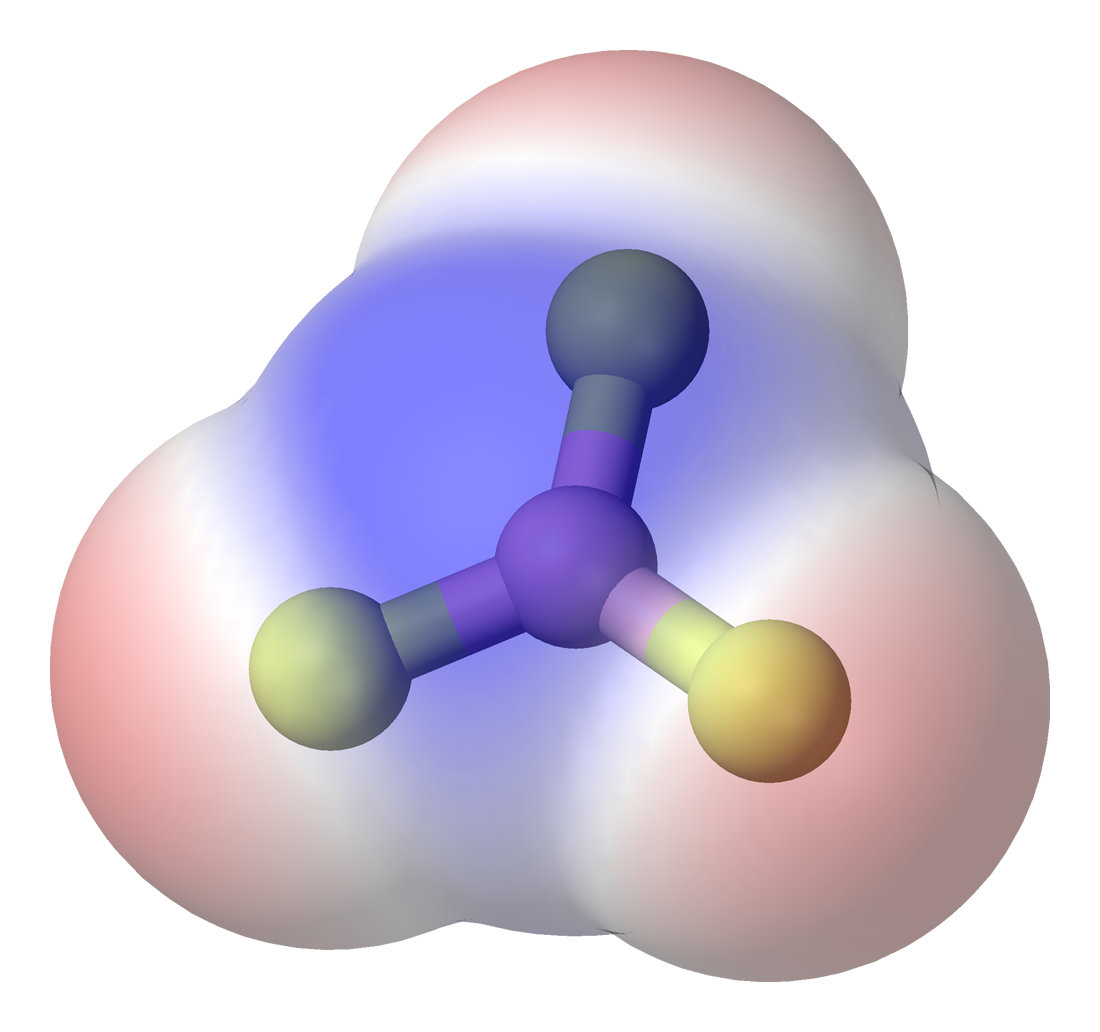 have a trigonal planar arrangement resulting in no overall dipole in the molecule, this is because symmetric shape allows the dipole moments from each \[\ce{B-F}\] bond to cancel each other out perfectly.
have a trigonal planar arrangement resulting in no overall dipole in the molecule, this is because symmetric shape allows the dipole moments from each \[\ce{B-F}\] bond to cancel each other out perfectly.
(Blue = partially positive region, Red = partially negative region)
Another example would be carbon dioxide, \[\ce{CO2}\], where it's linear \[\ce{O=C=O}\] shape allows the two bond dipole moments to cancel out each other resultin in no net molecular dipole moment.
Other examples of both polar/nonpolar molecules, molecules with a blue arrow signify a nonzero net dipole moment (essentially being a polar molecule):
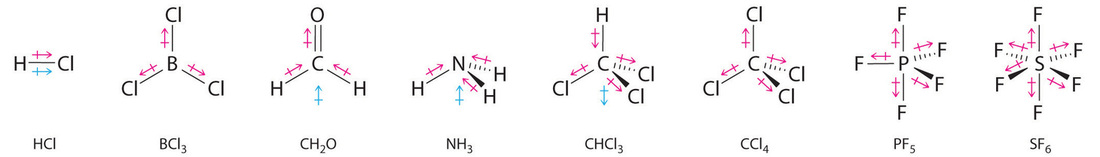
As it is shown here, although a polar bond is a prerequisite for a molecule to have a dipole, not all molecules with polar bonds exhibit dipoles. For \[\ce{AB_{n}}\] molecules, where \[\ce{A}\] is the central atom and \[\ce{B}\] are all the same types of atoms, there are certain molecular geometries which are symmetric. Therefore, they will have no dipole even if the bonds are polar. These geometries include linear, trigonal planar, tetrahedral, octahedral and trigonal bipyramid.
Properties of polar molecules
Boiling point
When comparing a polar and nonpolar molecule with similar molar masses, the polar molecule in general has a higher boiling point, because the dipole-dipole interactions between polar molecules results in stronger intermolecular attractions. One common form of polar interaction is the hydrogen bond.
Solubility
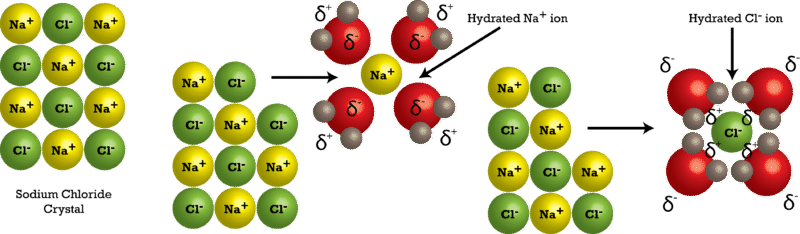
Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water. Most nonpolar molecules are water-insoluble (hydrophobic) at room temperature.
Another example would be glucose, (\[\ce{C6H12O6}\]). Since glucose has multiple \[\ce{-OH}\] groups:
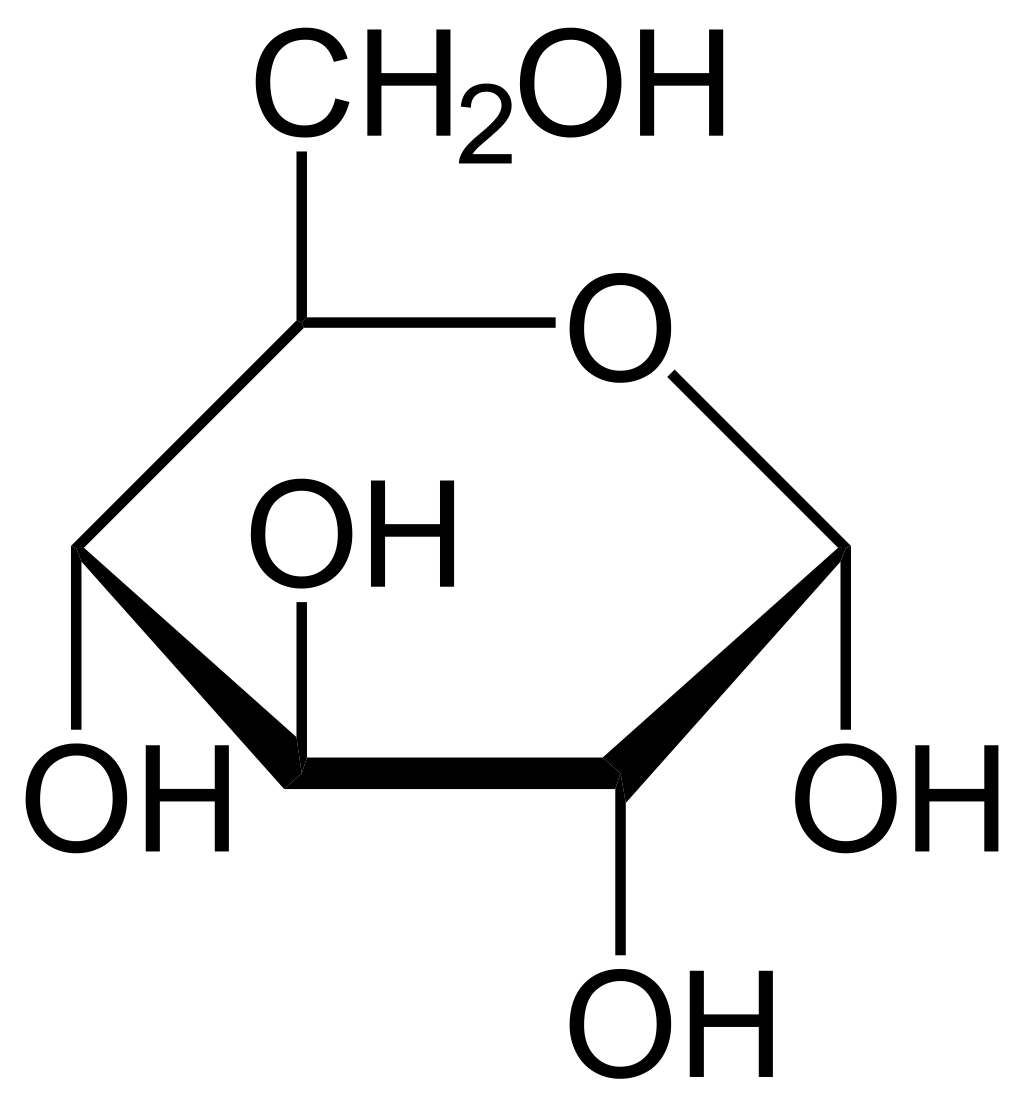
The polar hydroxyl groups in glucose can form hydrogen bonds with the water molecules. The oxygen of the hydroxyl group in glucose, having a partial negative charge, attracts the partially positive hydrogen of the water molecule. As glucose interacts with water, these attractions disrupt the intermolecular forces holding glucose molecules together in the solid state. The water molecules surround the glucose molecules, forming a hydration shell that stabilizes the glucose molecules in solution and prevents them from clumping back into a solid.
Viscosity
Polar liquids have a tendency to be more viscous than nonpolar liquids.