dipole
Dipole
A dipole is a separation of opposite electrical charges at close distances.
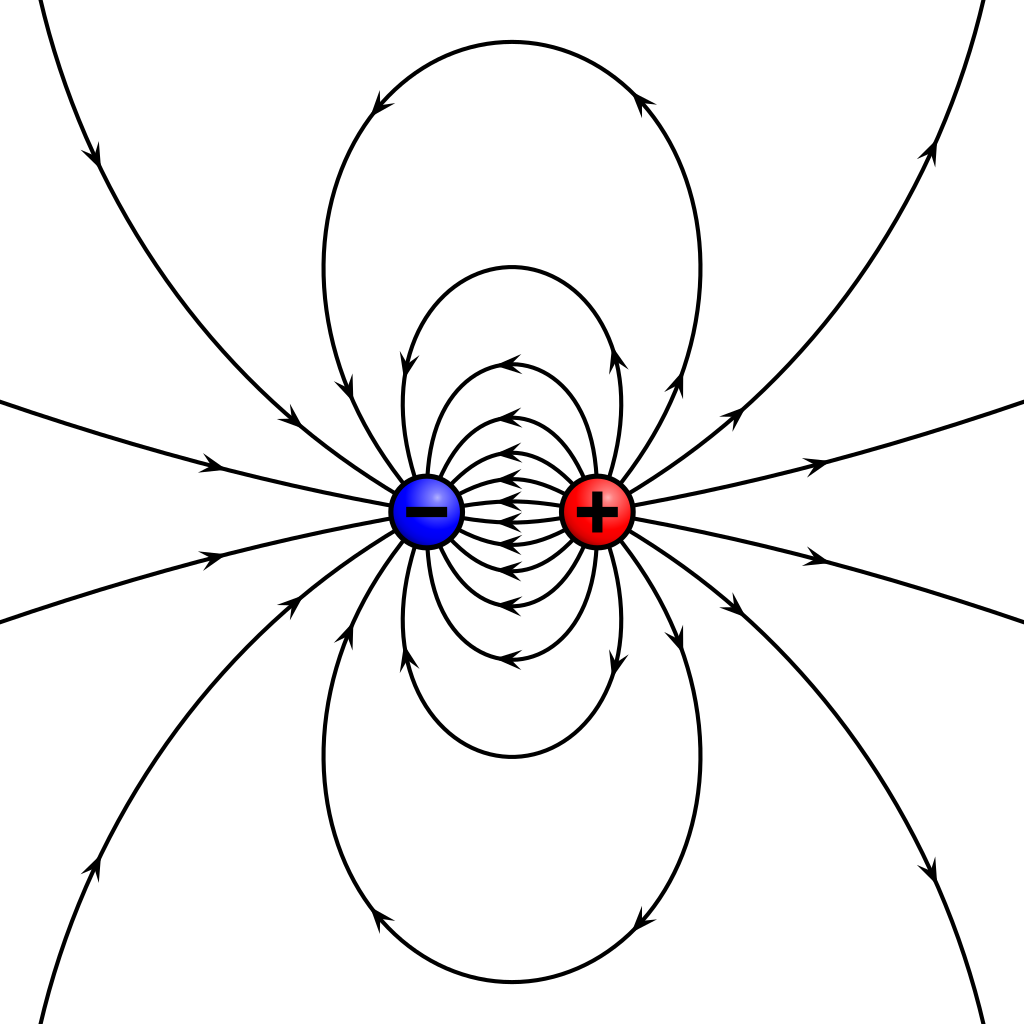
A dipole and dipole moment are quite different things. A dipole is anything (e.g. a molecule, even a part of a molecule) that has a non-zero dipole moment while a dipole moment is a mathematically clearly defined quantity, the product of charge difference and distance.
Electric dipoles
An electric dipole occurs when positive and negative charges (like a proton and an electron or a cation and an anion) are separate from each other. Usually, the charges are separated by a small distance.
Magnetic dipole
A magnetic dipole is the closed circulation of an electric current system. A simple example is a single loop of wire with constant current through it. A bar magnet is an example of a magnet with a permanent magnetic dipole moment.
Dipole moment
A dipole moment, \[\mu\], is a measurement of the separation of two opposite electrical charges. Dipole moments are a vector quantity. The magnitude is equal to the charge multiplied by the distance between the charges and the direction is from negative charge to positive charge. The magnitude of a single dipole moment is given by \[\mu=qd\], where \[q\] is the charge and \[d\] is the distance of separation.
For an array of charges (and if we also want to include the vector quantity), we use \[\overrightarrow{\mu}=\sum^{N}_{i=1}q_{i}\overrightarrow{d_{i}}\], where \[\overrightarrow{\mu}\] is the dipole moment vector, \[q_{i}\] is the magnitude of the \[i^{th}\] charge and \[\overrightarrow{d_{i}}\] is the vector representing the position of the \[i^{th}\] charge.
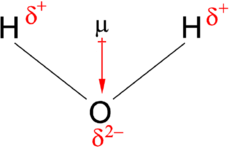
Sometimes the term "dipole" and "dipole moment" are used interchangeably, thus to avoid confusion, it's advised to used the term electric dipole moment, as per the recommendation of IUPAC.
Bond dipole moment
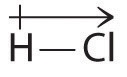
A bond dipole moment, \[\mu\], is the dipole moment of a single bond, e.g. the bond between \[\ce{H-Cl}\]. It is defined as the product of the partial charge \[Q\] on the bonded atoms and the distance \[d\] between the partial charges, and it is given by \[\mu=Qd\] where \[Q\] is measured in coulombs and \[d\] in meters.
We can measure the partial charges on the atoms in a molecule using the equation above. If the bonding were purely ionic, an electron would be transferred between the atoms, and there would be a full +1 charge on the donor and a full -1 charge on the acceptor. Now, let's say we were to calculate the bond dipole between \[\ce{H-Cl}\], assuming that \[\mu=1.109\text{D}\approx3.7\times10^{-30}\,\text{C m}\], where \[\text{D}\] represents debye and the gas phase \[\ce{H-Cl}\] distance is \[127.5\,\text{pm}\], the charge of each atom would be \[Q=\frac{3.7\times10^{-30}\,\text{C m}}{127.5\times10^{-12}\,\text{m}}=2.90\times10^{-20}\,\text{C}\]. Knowing the charge, we can simply convert it in terms of electrons, \[\frac{2.9\times10^{-20}\,\text{C}}{1.6022\times10^{-19}\,\text{C}}\approx0.181\,e^{-}\].
This tells us that the \[\ce{H-Cl}\] bond has approximately 18 percent ionic character or 82 percent covalent character. However \[0.181\,e^{-}\] does not mean a literal potion of electron is transferred, but rather the electron density being slightly skewed towards the chlorine atom. We can therefore indicate the charge separation quantitatively as \[\ce{\overset{0.181\delta^{+}}{H} - \overset{0.181\delta^{-}}{Cl}}\].