heterolysis
Heterolysis


Heterolysis or heterolytic fission (or heterolytic cleavage, if referring to the action) is the process of breaking a covalent bond where one previously bonded species takes both original bonding electrons from the other species. During heterolytic bond cleavage of a neutral molecule, a cation and an anion will be generated. Most commonly the more electronegative atom keeps the pair of electrons becoming anionic (negatively charged ion) while the more electropositive atom becomes cationic (positively charged ion).
Heterolysis almost always happens to single bonds; the process usually produces two fragment species. The energy required to break the bond is called the heterolytic bond-dissociation energy, which is similar (but not equivalent) to homolytic bond dissociation energy commonly used to represent the bond energy.
An arrow with the double-barbs is used to show heterolytic cleavage, that is the transfer of electron pair specifically.
Mechanism
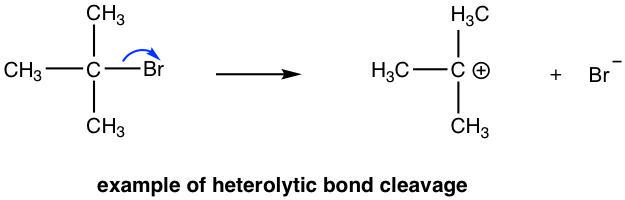
In the above example, the \[\ce{C-Br}\] bond is formed by the head-on (sigma) overlap of the sp3 hybridised orbitals from carbon and bromine. Given the electronegativities, the bonding molecular orbital formed is polarised towards bromine due to \[\ce{Br}\]'s slightly higher electronegativity thus creating a region of higher electron density around the bromine atom. When sufficient energy is supplied the electrons in this polarized bonding orbital are more readily transferred to bromine rather than being equally divided between carbon and bromine.
This electron transfer populates the antibonding orbitals minimally, meaning that while the bonding molecular orbital is being emptied of its electrons, the formation of any antibonding orbitals is kept to a minimum, thereby avoiding the energetically unfavorable occupation of antibonding states that would destabilize the bond further. Consequently, bromine retains both bonding electrons, resulting in the formation of a stable bromide ion, \[\ce{Br-}\] while the carbon is left electron deficient.