dipole-dipole interactions
Dipole-dipole interactions
Dipole-dipole interactions are electrostatic interactions between molecules which have permanent dipoles. This interaction is stronger than the London forces but is weaker than ion-ion interaction because only partial charges are involved. An example of a dipole-dipole interaction can be seen in hydrogen chloride (\[\ce{HCl}\]): the positive end of a polar molecule will attract the negative end of the other molecule and influence its position.
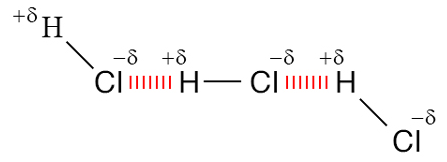
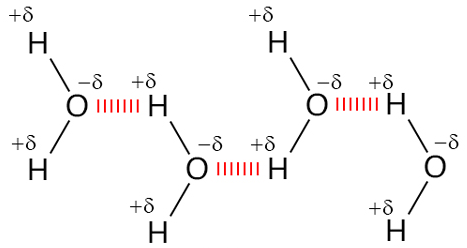
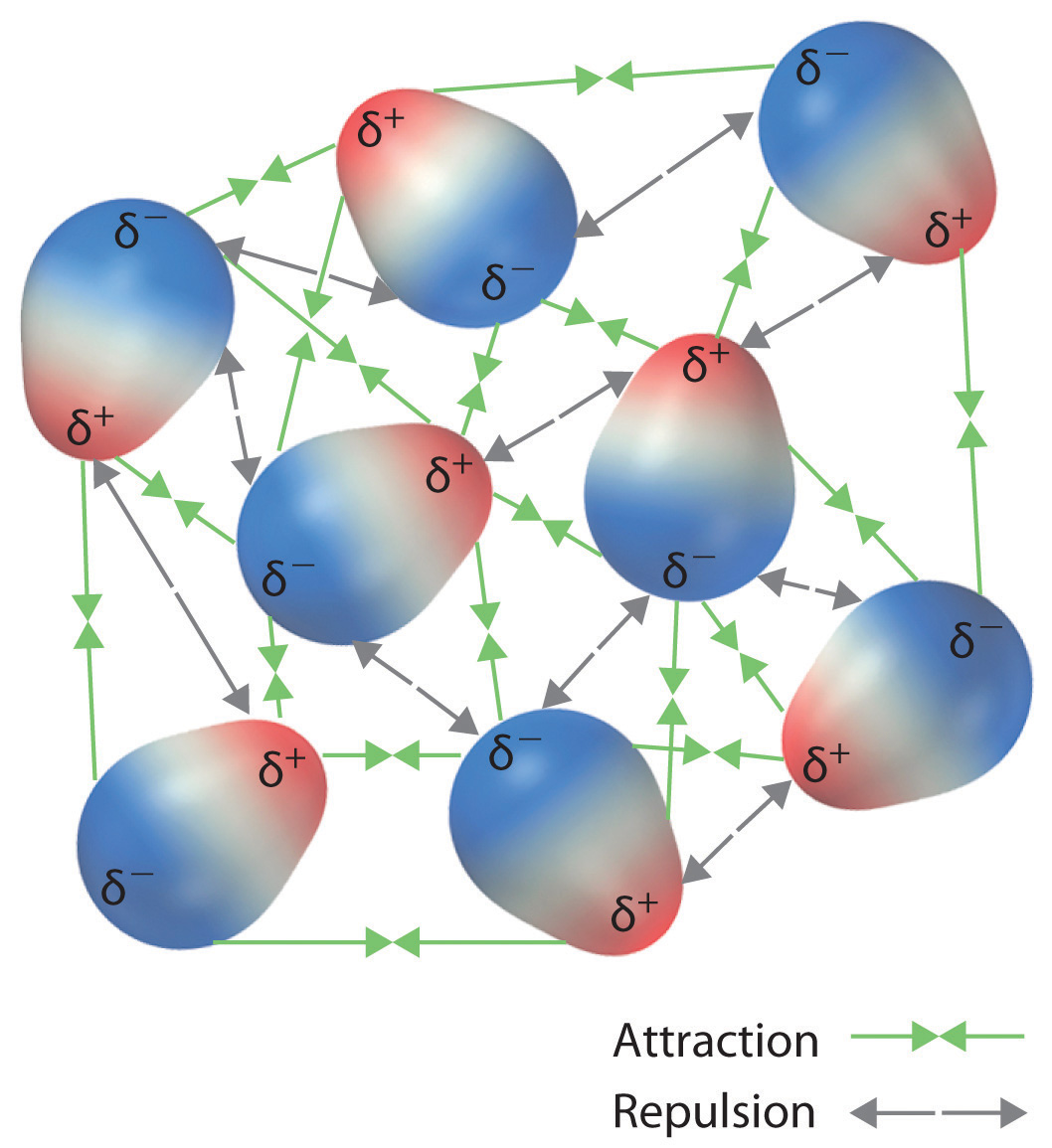
Although molecules in a liquid are in constant motion, they tend to align in the lowest energy orientation, which would be to maximize attractions and minimize repulsion.
Dipole-induced dipole interactions
A permanent dipole can induce a temporary dipole in a species that is normally nonpolar, and thus produce a net attractive force between the two particles When the two are brought together, the arrangement of electrons in the nonpolar molecule will be disrupted. As a result, it will acquire a partially positive charge and a partially negative charge. The two molecules will attract one another through dipole-induced dipole forces. It is a weaker interaction than the regular dipole-dipole interaction.
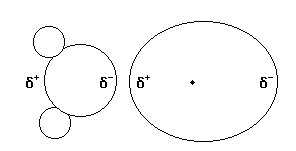
In the above scenario, the polar water molecule has induced a dipole in the nonpolar argon atom.