ionic bond
Ionic bonding
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. Atoms that gain electrons make negatively charged ions (called anions) while atoms that lose electrons make positively charged ions (called cations). Ionic bond is an electrostatic force between positive and negative ions. This electrostatic force in non-directional, it acts in all directions. The strength of the force depends on the magnitude of the charges on the ions and the sum of the ionic radii.
Purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing electron density between them. Therefore, all ionic bonding has some covalent character. Thus, bonding is considered ionic where the ionic character is greater than the covalent character. The larger the difference in electronegativity between the two types of atoms involved in the bonding, the more ionic (polar) it is.
Calcium floride \[\ce{CaF2}\], is an ionic compound.
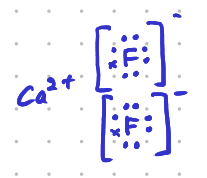
Forces
The forces holding ions together in ionic solids are electrostatic forces (Coulomb force). Opposite charges attract each other. These are the strongest intermolecular forces. Ionic forces hold many ions in a crystal lattice structure.
Properties
-
Ionic bonds are considered to be among the strongest of all types of chemical bonds. This often causes ionic compounds to be very stable.
Ionic bonds are stronger usually stronger than covalent bonds as in a fully ionic bond you have a coulomb interaction with opposite charges. These two ions want to be as close together as possible, resulting in a stronger bond. Generally speaking, Coulomb interactions have a greater impact than molecular orbitals on the strength of a bond.
- Ionic bonds have high bond energy.
-
Most ionic compounds exist in the form of a crystal structure, in which the ions occupy the corners of the crystal. Such a structure is called a crystal lattice.

Crystal structure of table salt (sodium in purple, chlorine in green).
- Ionic compounds lose their crystal lattice structure and break up into ions when dissolved in water or any other polar solvent. The presence of these free ions makes aqueous ionic compound solutions good conductors of electricity. The same occurs when the compounds are heated above their melting point and melted.
Electron density
Electron density of compound \[\ce{NaCl}\]:
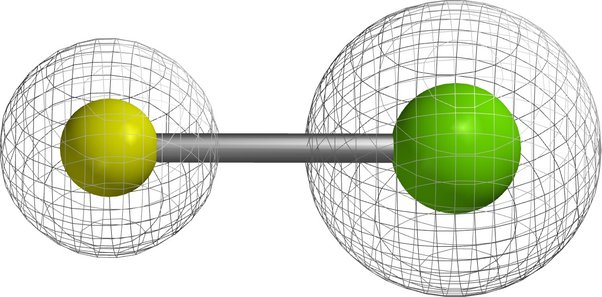
In ionic compounds like NaCl, ions typically display a spherical electron density. This is because the electron density around an ion in an ionic compound is more evenly distributed, as ions are often in their most stable (closed shell) electronic configuration after electron transfer. The description of sodium (\[\ce{Na}\]) as having a more contracted electron density reflects the loss of an electron (forming \[\ce{Na+}\]), while chlorine (\[\ce{Cl}\]) having a more expanded electron density due to gaining an electron (forming \[\ce{Cl-}\]) .
The attractive force exerted by an ion does not favor any particular direction relative to its orientation, which is a fundamental characteristic of ionic bonding. The structure of ionic solids depends primarily on the size and ratios of the ions that make up the crystal lattice. This influences how the ions pack together, often in a highly ordered, repeating pattern that maximizes the stability of the solid. This contrasts with organic materials, where directed bonds (like the covalent bond) and the geometry of molecular orbitals play a more significant role in determining structure.