molecular structure
Molecular structures
A bond angle given is the ideal geometric angle (may differ for different molecules usually from \[\pm2^{\circ}\] to \[\pm5^{\circ}\]) between two adjacent bonds. Key: White = peripheral atom, Yellow = lone pair electron, ax = axial ligand, eq = equatorial ligand (see trigonal bipyramidal).
See molecular shape diagram, AXE notation from VSEPR theory, coordination number and orbital hybridisation. Do note that the orbital hybridisation for shapes with lone pairs will be inaccurate, especially for hybridised orbitals containing the d orbital.
The relation between the different shapes and the hybridised orbitals is that the bonds between the ligands and central atom are hybridised orbitals.
Linear
The linear molecular geometry describes the geometry around a central atom bonded to two other atoms (or ligands) placed at a bond angle of \[180^{\circ}\].
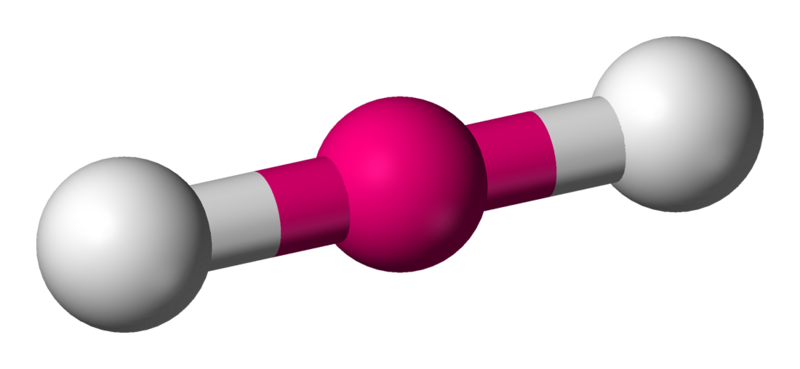
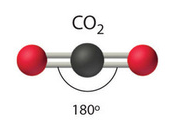 Coordination number: 2, sp, \[\ce{AX_{2}E_{0}}\].
Coordination number: 2, sp, \[\ce{AX_{2}E_{0}}\].
For molecules such as \[\ce{CO2}\] there are no lone pairs in the center atom, therefore has a linear shape.
Another situation where a molecule has a linear structure is \[\ce{XeF2}\]:
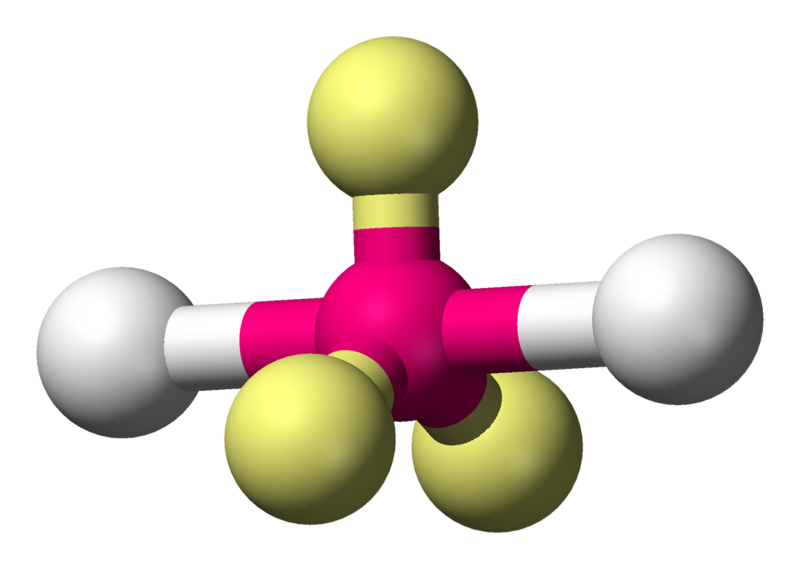 Coordination number: 2, \[\ce{AX_{2}E_{3}}\]
Coordination number: 2, \[\ce{AX_{2}E_{3}}\]
Trigonal planar
A model with one atom at the center and three atoms at the corners of an equilateral triangle, called peripheral atoms, all in one plane. In an ideal trigonal planar species, all three ligands are identical and all bond angles are \[120^{\circ}\].
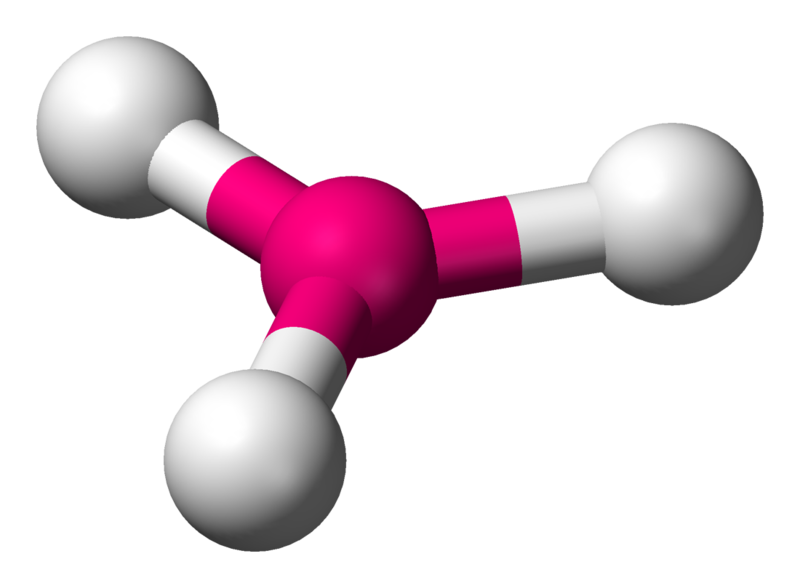
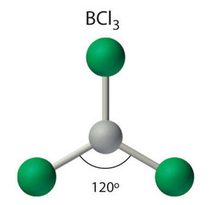 Coordination number: 3, \[\ce{sp^{2}}\], \[\ce{AX_{3}E_{0}}\]
Coordination number: 3, \[\ce{sp^{2}}\], \[\ce{AX_{3}E_{0}}\]
Bent
olecules with a non-collinear arrangement of two adjacent bonds have bent molecular geometry. Certain atoms, such as oxygen, will almost always set their two (or more) covalent bonds in non-collinear directions due to their electron configuration. There are several variants of bending, where the most common is \[\ce{AX_{2}E_{2}}\] where two covalent bonds and two lone pairs of the central atom form a complete 8-electron shell. They have central angles from \[104^{\circ}\] to \[109.5^{\circ}\]. The most common actual angles are \[105^{\circ}\], \[107^{\circ}\], and \[109^{\circ}\]: they vary because of the different properties of the peripheral atoms. \[\ce{AX_{2}E_{1}}\] molecules, such as \[\ce{SnCl2}\], have only one lone pair and the central angle about \[120^{\circ}\].
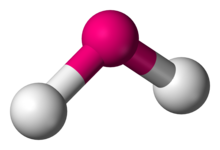
Range of possible bond angles: \[90^{\circ}<\theta<120^{\circ}\]
A molecule would have such a shape due to the VSEPR theory, where it states that there exists lone pair-bonding pair repulsion as shown below:
 \[\ce{SO2}\]:
\[\ce{SO2}\]: 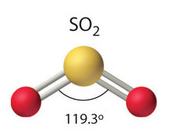 Coordination number: 2, \[\ce{sp^{2}}\], \[\ce{AX_{2}E_{1}}\]
Coordination number: 2, \[\ce{sp^{2}}\], \[\ce{AX_{2}E_{1}}\]
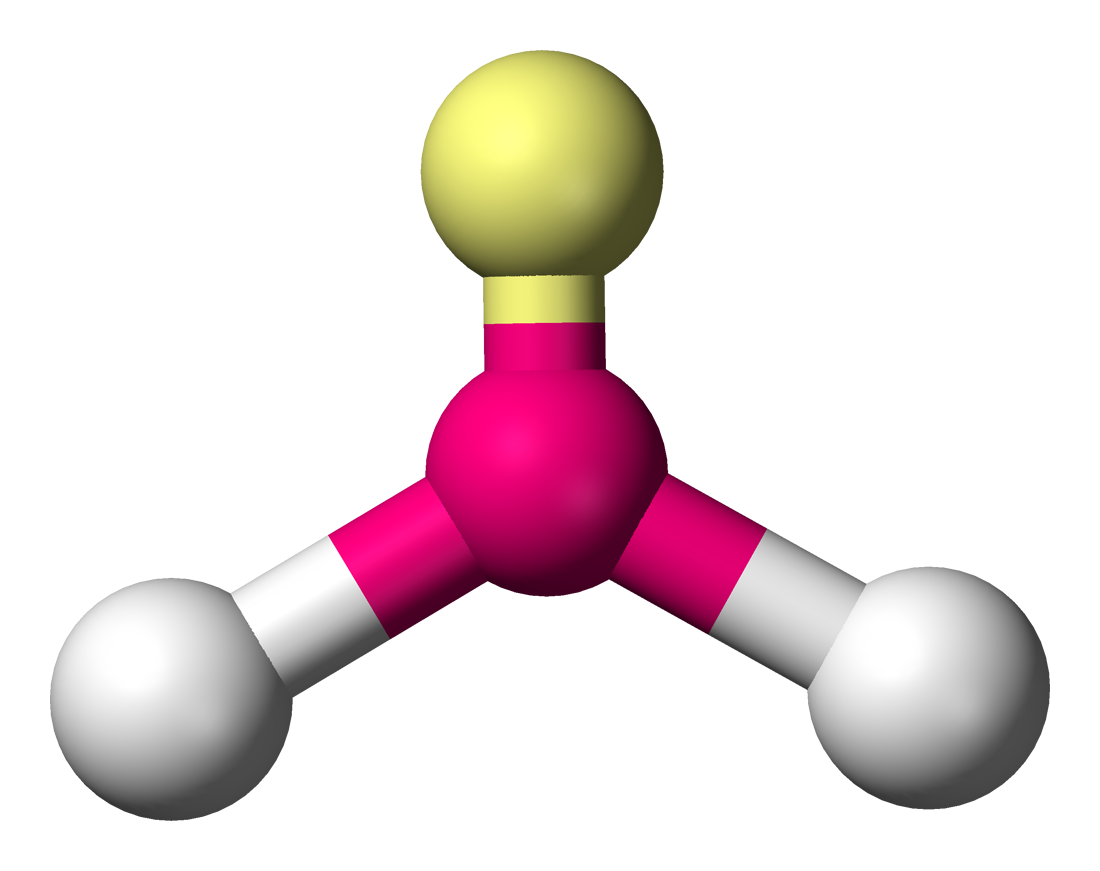
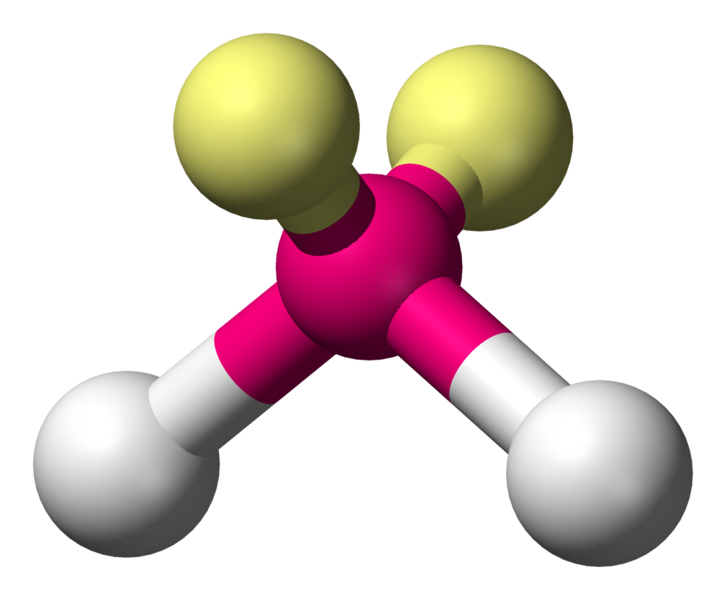 \[\ce{H2O}\]:
\[\ce{H2O}\]:  Coordination number: 2, \[\ce{sp^{3}}\], \[\ce{AX_{2}E_{2}}\]
Coordination number: 2, \[\ce{sp^{3}}\], \[\ce{AX_{2}E_{2}}\]
Tetrahedral
A central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are \[\cos^{-1}\left(-\frac{1}{2}\right)=109.4712^{\circ}\approx109.5^{{\circ}}\] when all four substituents are the same, as in methane (\[\ce{CH4}\]).
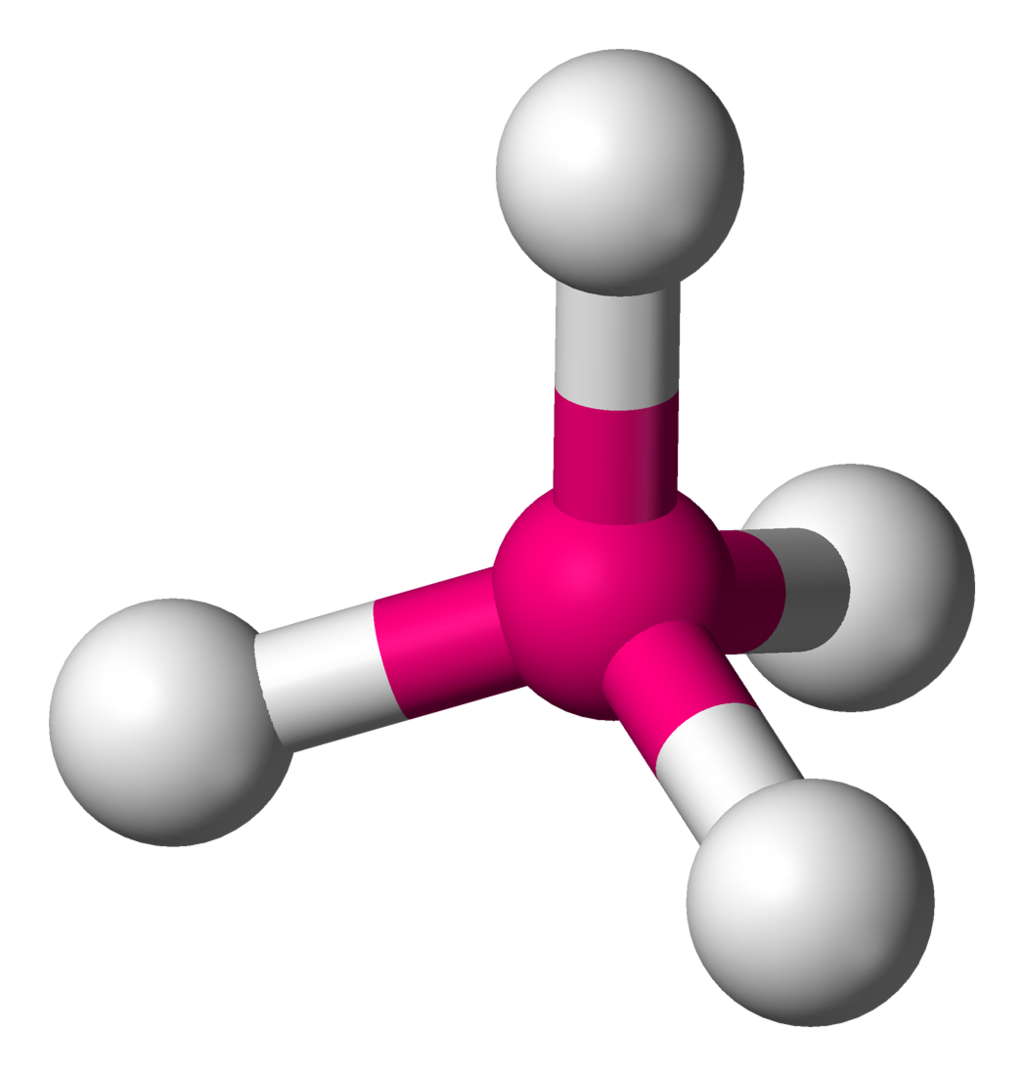 \[\ce{CH4}\]:
\[\ce{CH4}\]: 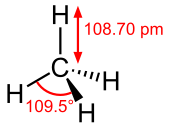 Coordination number: 4, \[\ce{sp^{3}}\], \[\ce{AX_{4}E_{0}}\]
Coordination number: 4, \[\ce{sp^{3}}\], \[\ce{AX_{4}E_{0}}\]
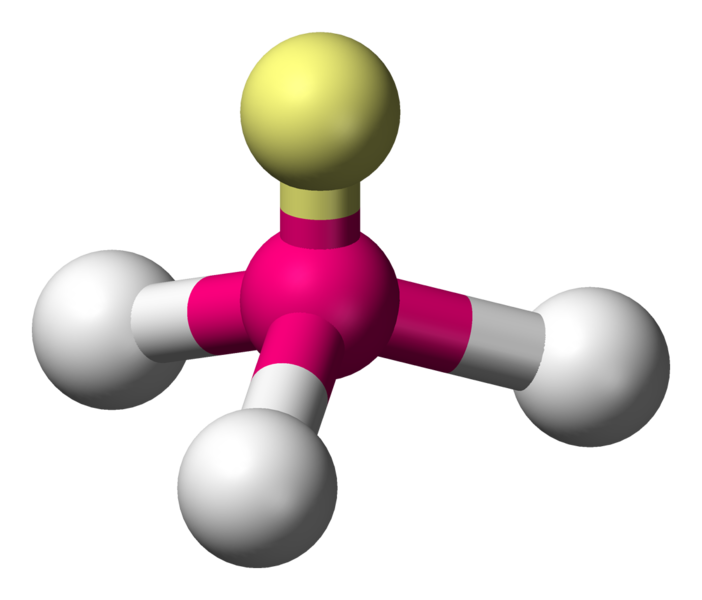
Trigonal pyramidal
One atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron. Ideally the bond angles would be also \[109.5^{\circ}\], but a case such as the nitrogen in ammonia (\[\ce{NH3}\]) which has 5 valence electrons and bonds with three hydrogen atoms begs to differ. The three hydrogen atoms are repelled by the electron lone pair in a way that the actual bond angles are \[106.7^{\circ}\].
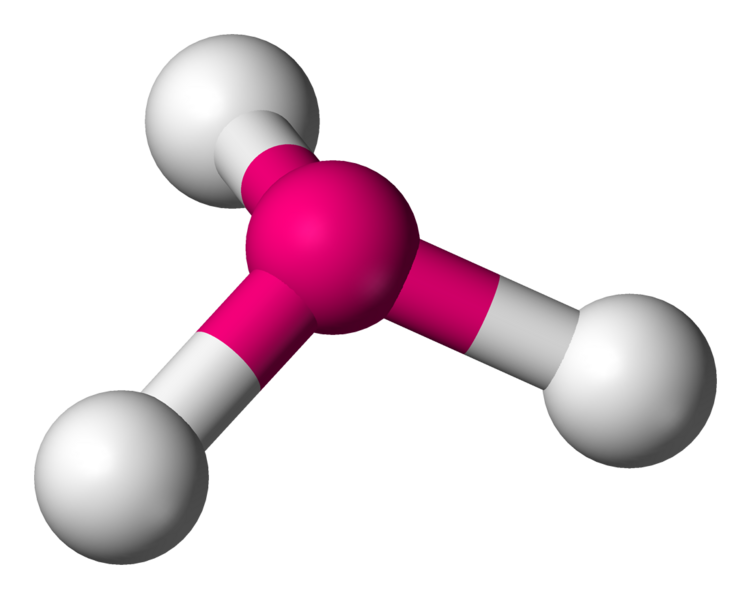
\[\ce{NH3}\]:
 due to the
due to the 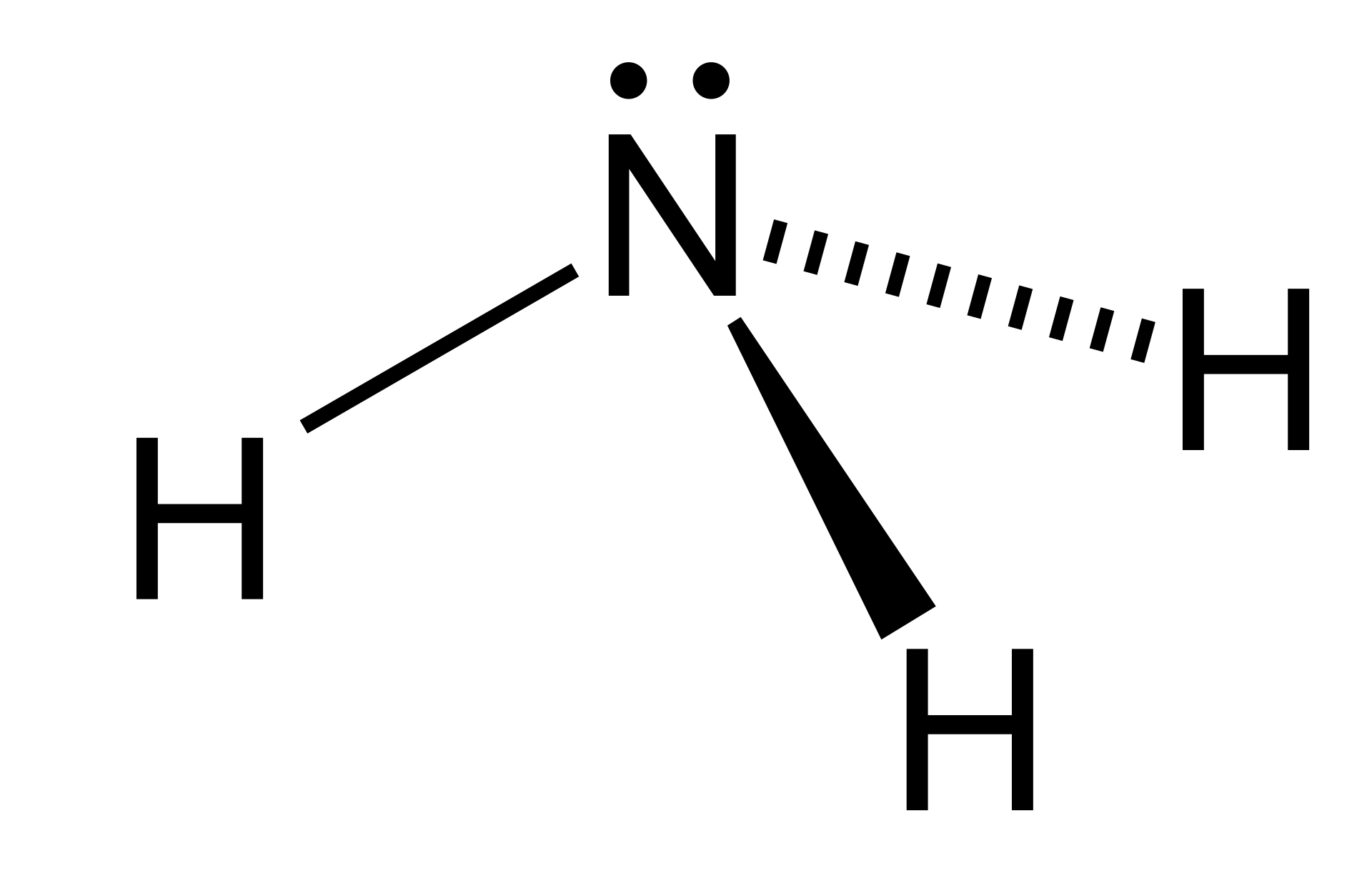 or
or 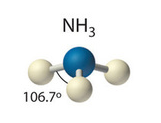 Coordination number: 3, \[\ce{sp^{3}}\], \[\ce{AX_{3}E_{1}}\]
Coordination number: 3, \[\ce{sp^{3}}\], \[\ce{AX_{3}E_{1}}\]
Trigonal bipyramidal
One atom at the center and 5 more atoms at the corners of a triangular bipyramid. This is one geometry for which the bond angles surrounding the central atom are not identical, because there is no geometrical arrangement with five terminal atoms in equivalent positions. Examples of this molecular geometry are phosphorus pentafluoride (\[\ce{PF5}\]). The ideal bond angle between the eq and ax (perpendicular to each other) would be \[90^{\circ}\] while between eq and eq would be \[120^{\circ}\].
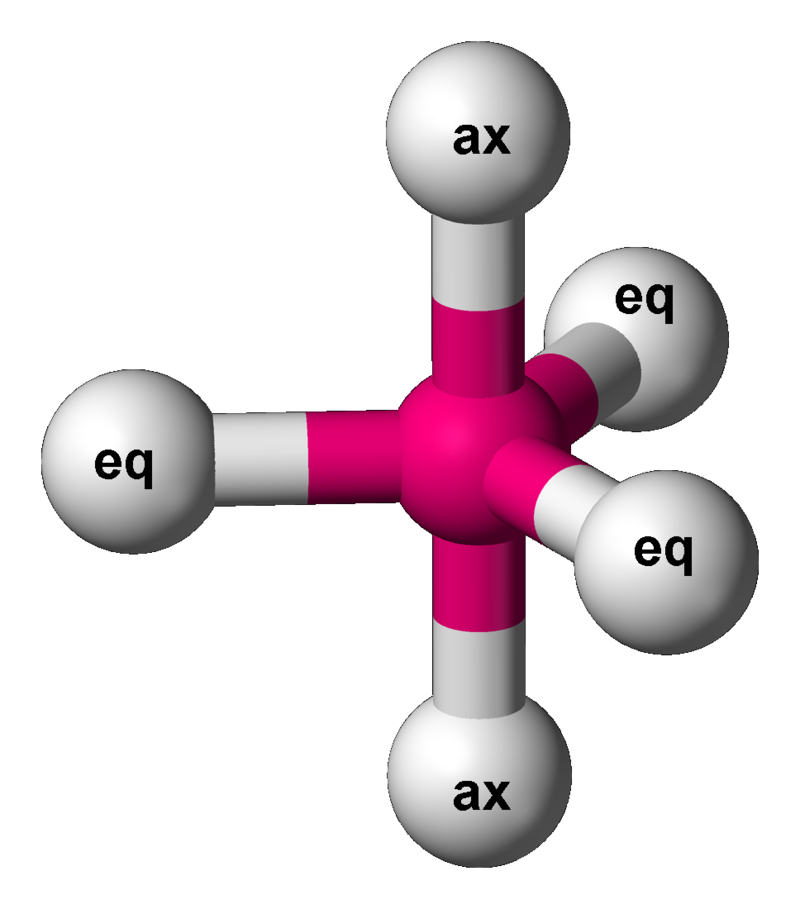 \[\ce{PF5}\]:
\[\ce{PF5}\]: 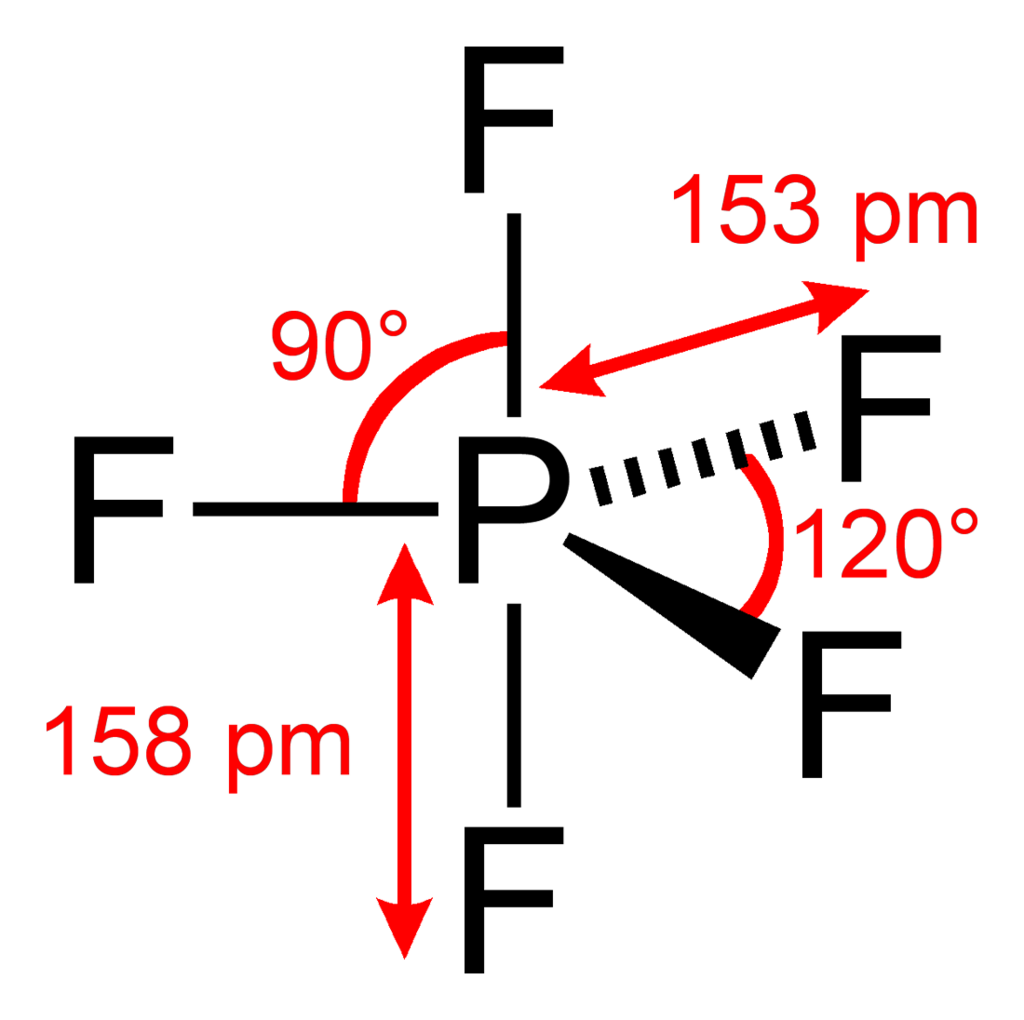 Coordination number: 5, \[\ce{sp^{3}d}\], \[\ce{AX_{5}E_{0}}\]
Coordination number: 5, \[\ce{sp^{3}d}\], \[\ce{AX_{5}E_{0}}\]
Seesaw
Four bonds to a central atom. Similar to the trigonal bipyramid with two axial and three equatorial positions, but in the seesaw geometry one of the atoms is replaced by a lone pair of electrons, which is always in an equatorial position. This is true because the lone pair occupies more space near the central atom than does a bonding pair of electrons.
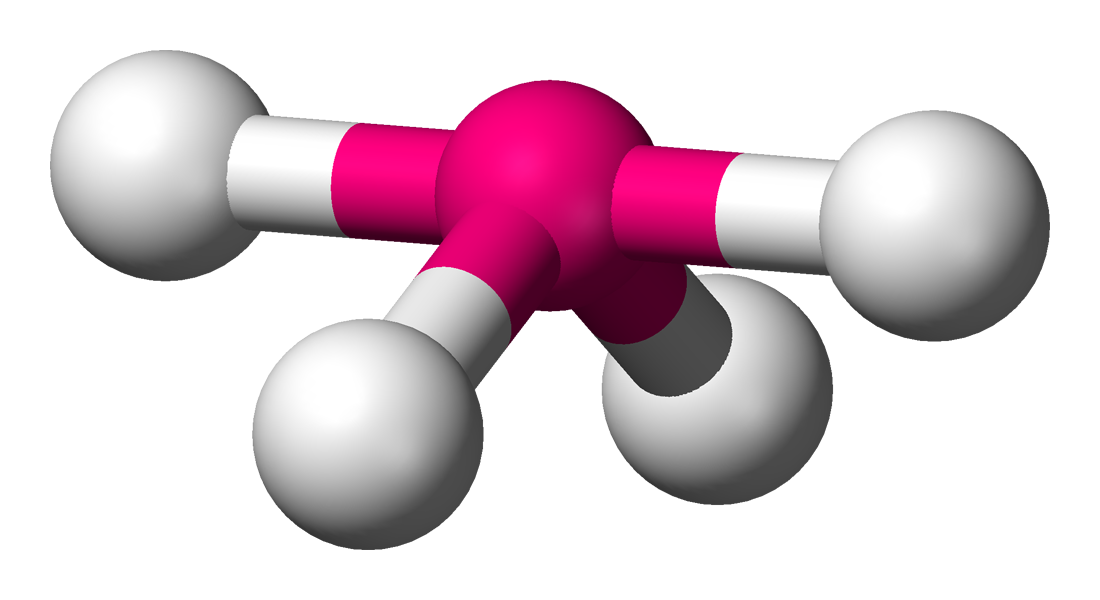
The ideal bond angles above would be \[180^{\circ}\] at ax-ax (as the lone pair is in eq position), eq-eq at \[120^{\circ}\] and ax-eq at \[90^{\circ}\].
With the lone pair:
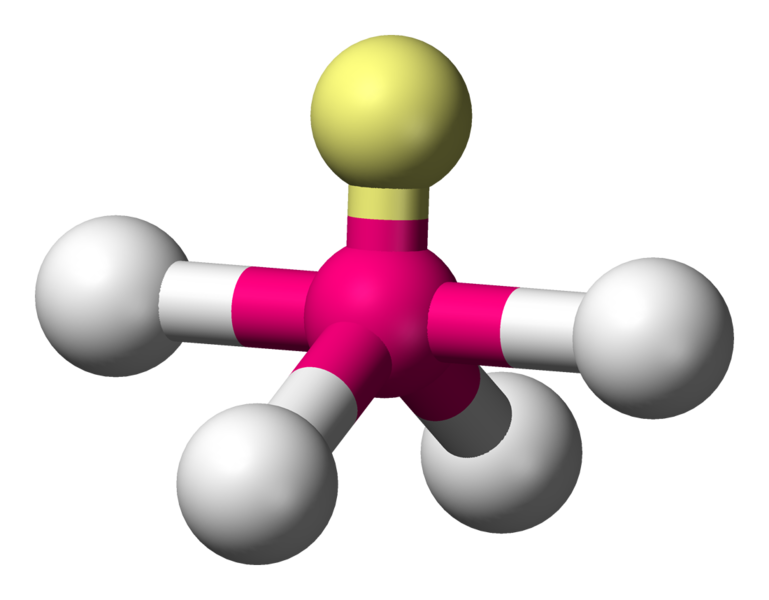 \[\ce{SF4}\]:
\[\ce{SF4}\]: 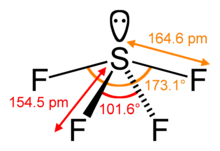 Coordination number: 4, \[\ce{sp^{3}d}\], \[\ce{AX_{4}E_{1}}\]
Coordination number: 4, \[\ce{sp^{3}d}\], \[\ce{AX_{4}E_{1}}\]
T-shaped
Describes the structures of some molecules where a central atom has three ligands. According to VSEPR theory, T-shaped geometry results when three ligands and two lone pairs of electrons are bonded to the central atom.
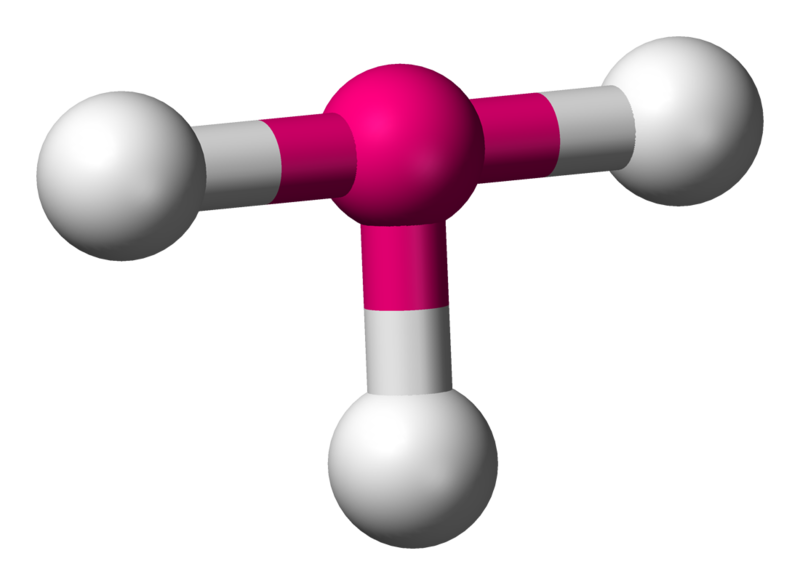
Ideally, the bond angles would be at \[90^{\circ}\] and \[180^{\circ}\], but again, lone pair repulsion > bonding pair repulsion.
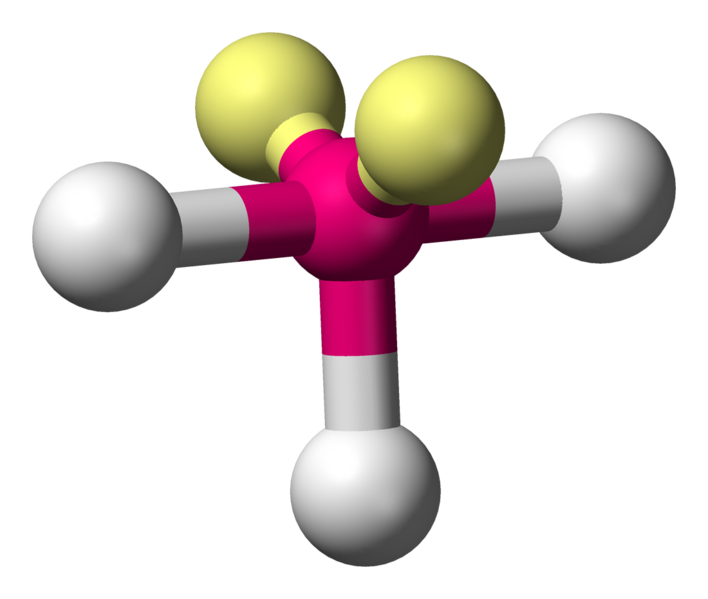 \[\ce{ClF3}\]:
\[\ce{ClF3}\]: 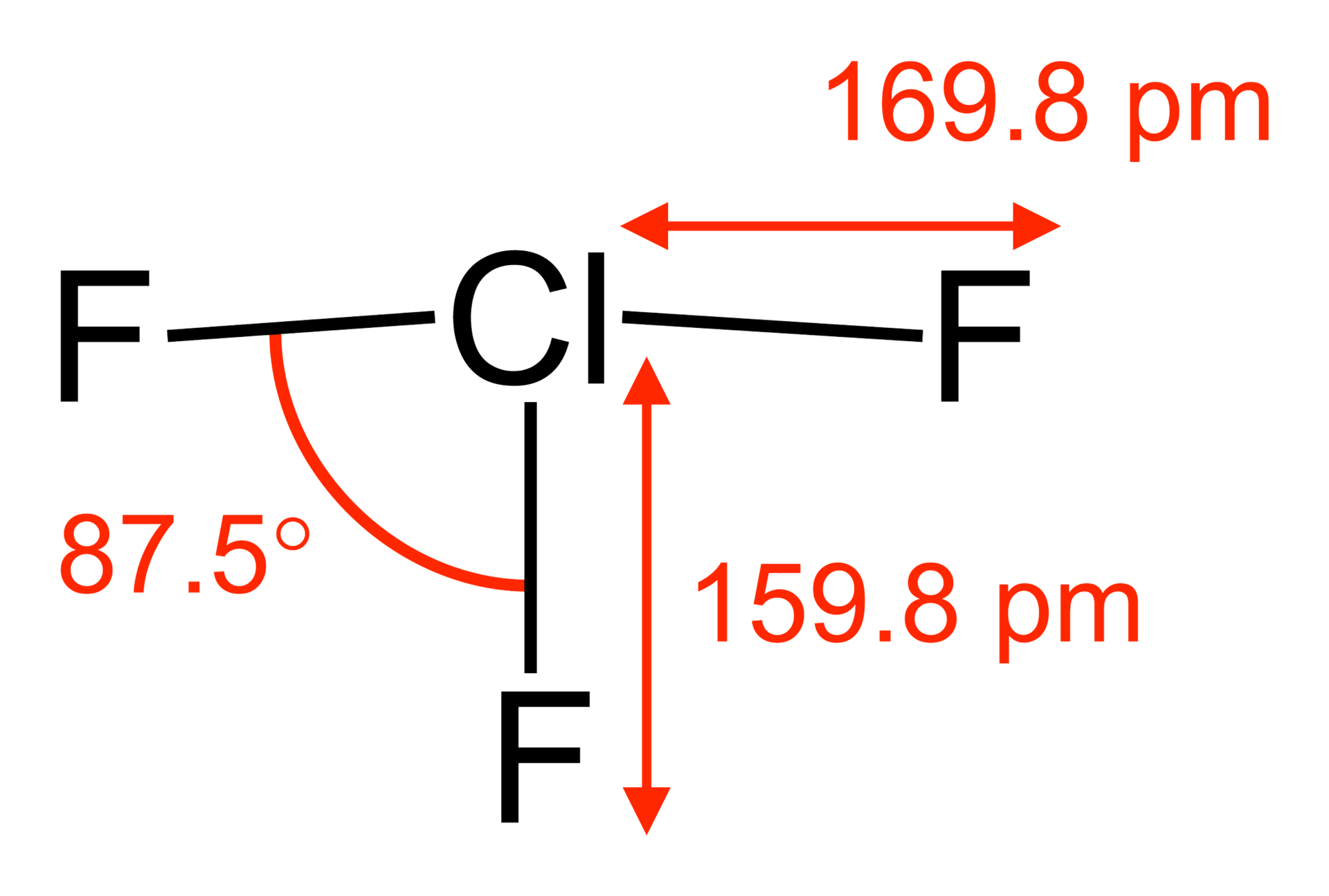 Coordination number: 3, \[\ce{sp^{3}d}\], \[\ce{AX_{3}E_{2}}\]
Coordination number: 3, \[\ce{sp^{3}d}\], \[\ce{AX_{3}E_{2}}\]
Square bipyramidal/Octahedral
Six atoms or groups of atoms or ligands symmetrically arranged around a central atom, defining the vertices of an octahedron.
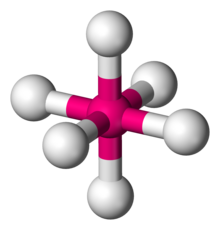 \[\ce{SF6}\]:
\[\ce{SF6}\]: 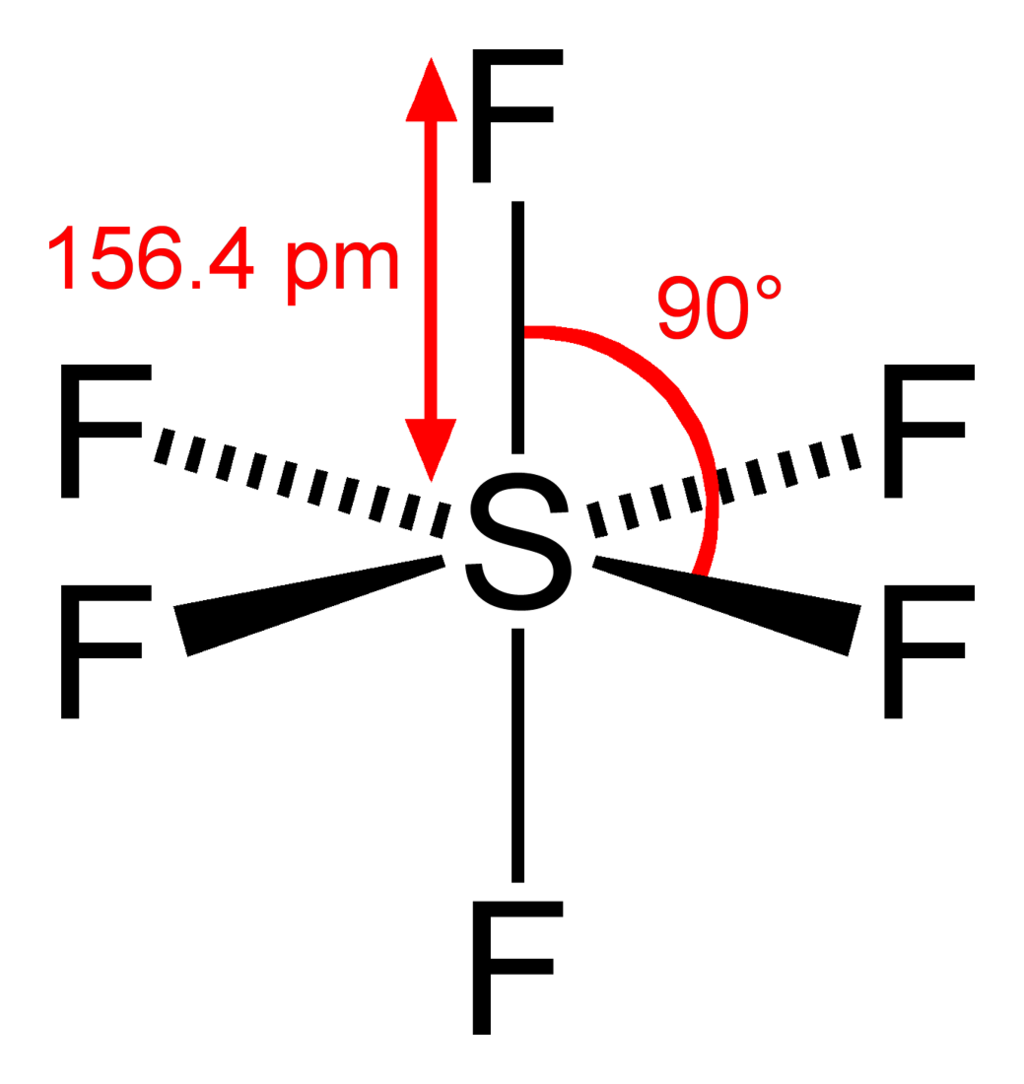 Coordination number: 6, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{6}E_{0}}\]
Coordination number: 6, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{6}E_{0}}\]
The bond angles here would be \[90^{\circ}\] and \[180^{\circ}\].
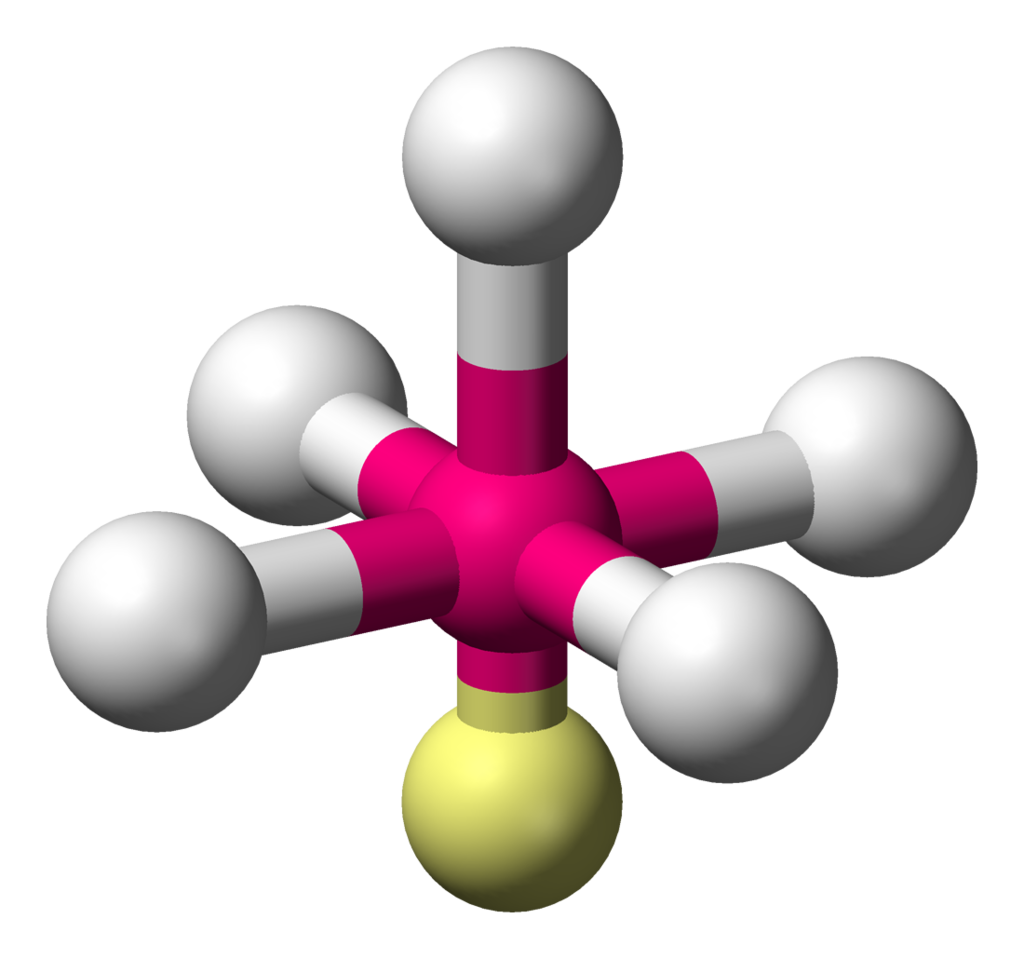
Square pyramidal
Describes the shape of certain chemical compounds with the formula \[\ce{ML5}\] where \[\ce{L}\] is a ligand.
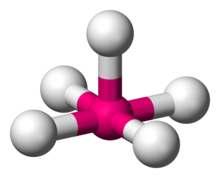
The bond angles would be ideally \[90^{\circ}\].
 \[\ce{ClF5}\]:
\[\ce{ClF5}\]: 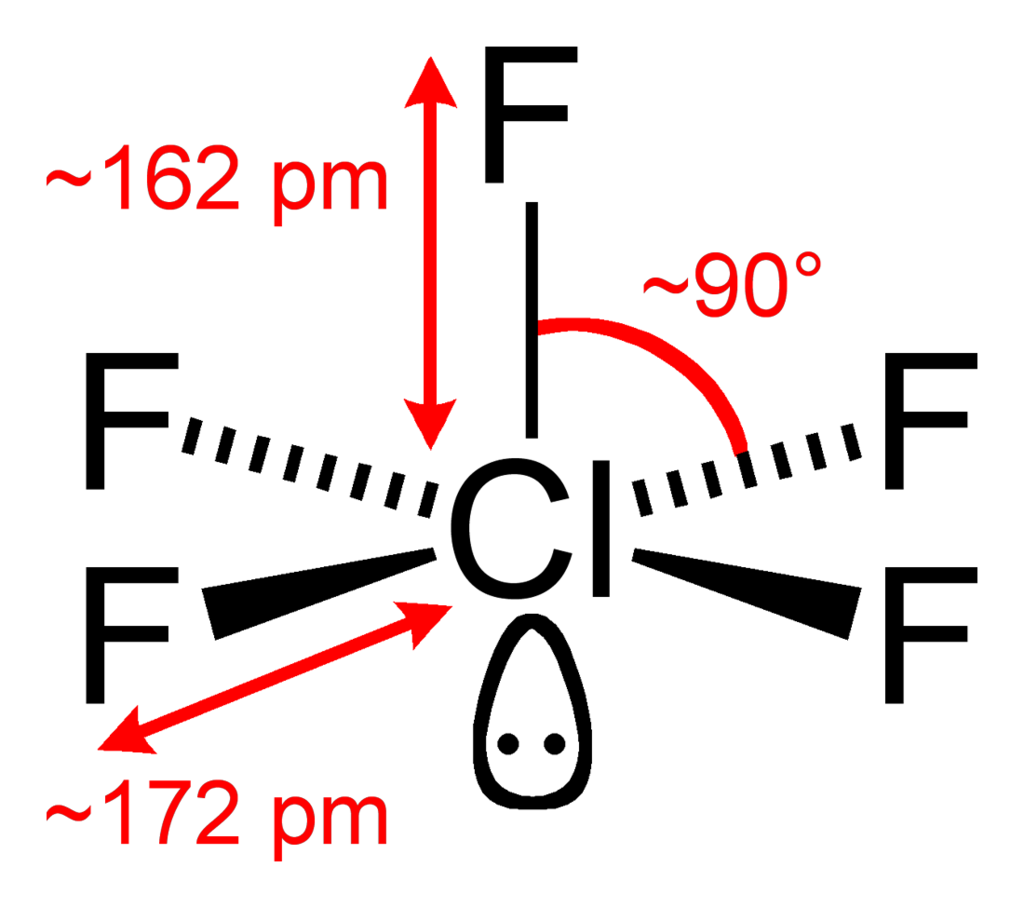 Coordination number: 5, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{5}E_{1}}\]
Coordination number: 5, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{5}E_{1}}\]
Square planar
Numerous compounds adopt this geometry, especially for transition metal complexes. An example of such a compound is \[\ce{XeF4}\].
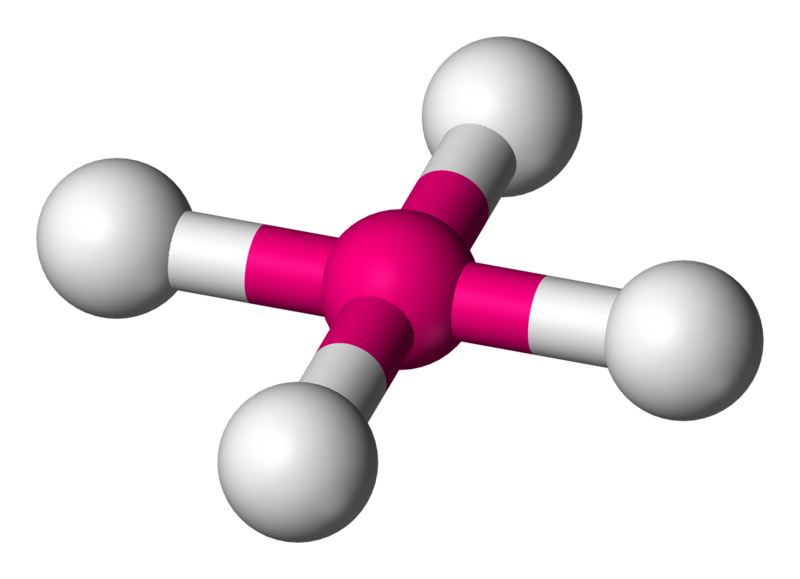
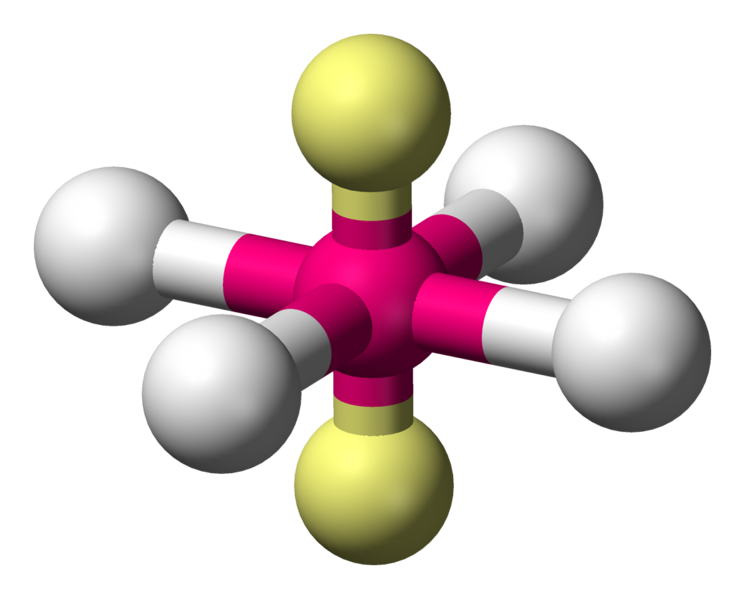 Coordination number: 4, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{4}E_{2}}\]
Coordination number: 4, \[\ce{sp^{3}d^{2}}\], \[\ce{AX_{4}E_{2}}\]
The bond angles would be ideally \[90^{\circ}\] and \[180^{\circ}\].
Pentagonal bipyramidal
A molecular geometry with one atom at the centre with seven ligands at the corners of a pentagonal bipyramid.
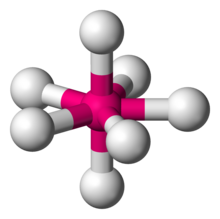 \[\ce{IF7}\]:
\[\ce{IF7}\]: 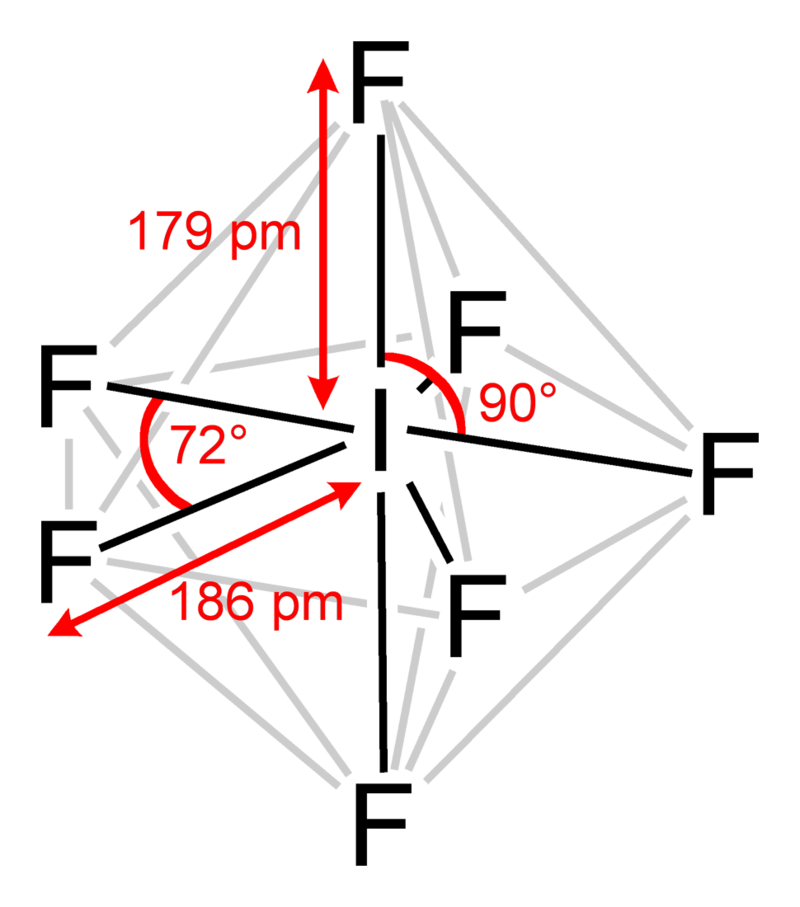 Coordination number: 7, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{7}E_{0}}\]
Coordination number: 7, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{7}E_{0}}\]
\[72^{\circ}\] and \[90^{\circ}\] bond angles.
Pentagonal pyramidal
\[\ce{XeOF5-}\]
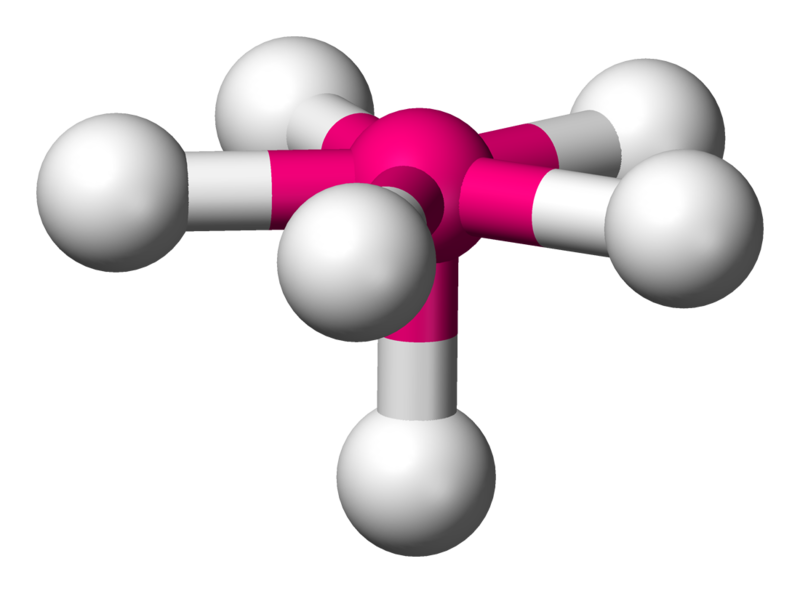
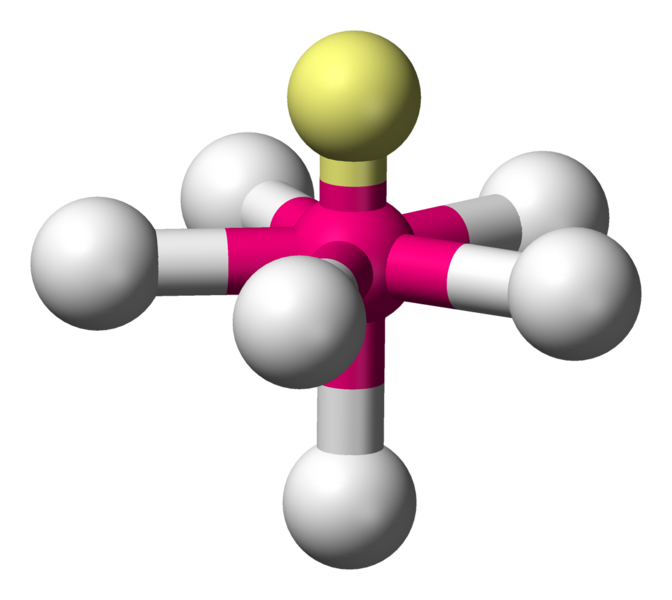 Coordination number: 6, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{6}E_{1}}\]
Coordination number: 6, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{6}E_{1}}\]
\[72^{\circ}\], \[90^{\circ}\] and \[144^{\circ}\] bond angles.
Pentagonal planar
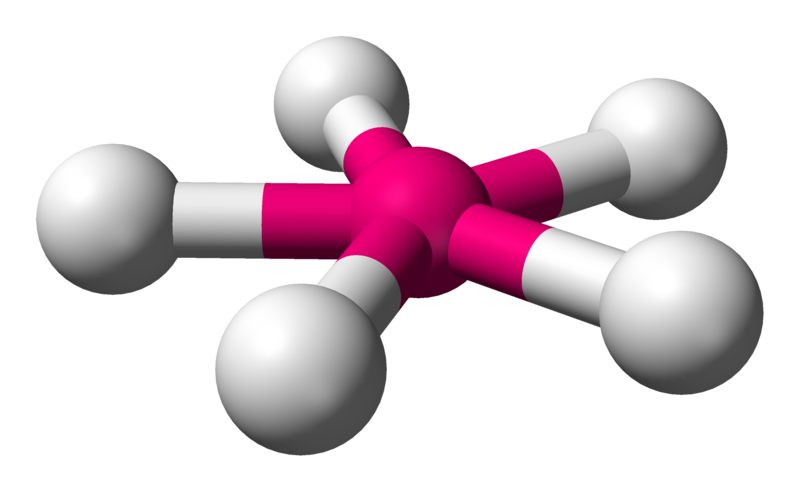
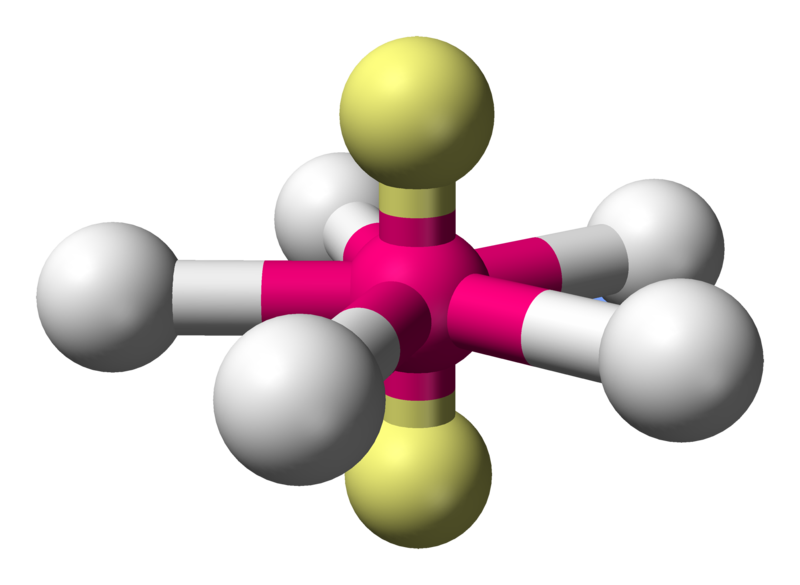 \[\ce{XeF5-}\]:
\[\ce{XeF5-}\]:  Coordination number: 5, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{5}E_{0}}\]
Coordination number: 5, \[\ce{sp^{3}d^{3}}\], \[\ce{AX_{5}E_{0}}\]
\[72^{\circ}\] and \[144^{\circ}\] bond angles.