crystalline solids
Crystalline solids
Ionic crystals
Ionic crystals, or giant ionic structures, such as sodium chloride and nickel oxide, are composed of positive and negative ions that are held together by electrostatic attractions (Coulomb force), which can be quite strong. Many ionic crystals also have high melting points. This is due to the very strong attractions between the ions in ionic compounds, as the attractions between full charges are (much) larger than those between the partial charges in polar molecular compounds. Although they are hard, they also tend to be brittle, and they shatter rather than bend. Ionic solids do not conduct electricity; however, they do conduct when molten or dissolved because their ions are now free to move.
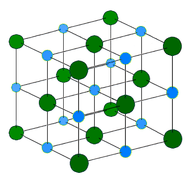
Metallic solid
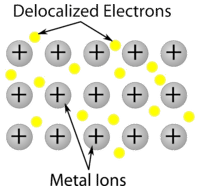
Metallic solids, or giant metallic structures, such as crystals of copper, aluminum, and iron are formed by metal atoms. The structure of metallic crystals is often described as a uniform distribution of atomic nuclei within a “sea” of delocalized electrons. The atoms
within such a metallic solid are held together by metallic bonding that gives rise to many useful and varied bulk properties. All exhibit high thermal and electrical conductivity, metallic luster, and malleability.
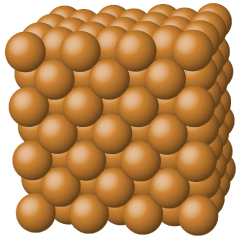
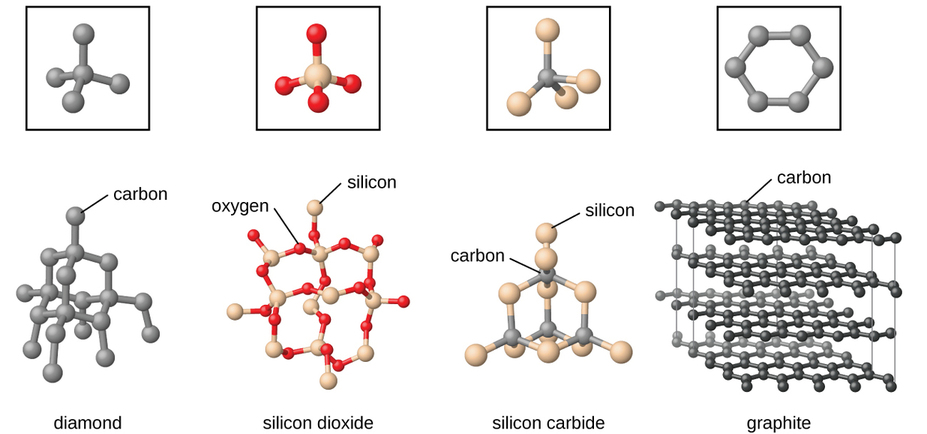
Covalent network solids
Also known as giant covalent/molecular structures is a chemical compound (or element) in which the atoms are bonded by covalent bonds in a continuous network extending throughout the material. In a network solid there are no individual molecules, and the entire crystal may be considered a macromolecule. To break or to melt a covalent network solid, covalent bonds must be broken. Because covalent bonds are relatively strong, covalent network solids are typically characterized by hardness, strength, and high melting points.
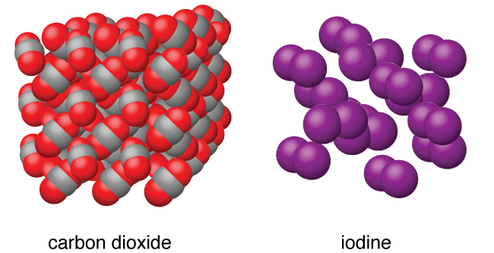
Molecular solids
A molecular solid, or simple molecular, is a solid consisting of discrete molecules. The cohesive forces that bind the molecules together are van der Waals forces, dipole-dipole interactions, pi bond-pi bond interactions, hydrogen bonding, London dispersion forces, and in some molecular solids, Coulombic interactions.
The strengths of the attractive forces between the units present in different crystals vary widely, as indicated by the melting points of the crystals. Small symmetrical molecules (nonpolar molecules), such as \[\ce{H2}\], \[\ce{N2}\], \[\ce{O2}\], and \[\ce{F2}\], have weak attractive forces and form molecular solids with very low melting points (below -200C). Substances consisting of larger, nonpolar molecules have larger attractive forces and melt at higher temperatures.