nucleophilic addition
Nucleophilic addition
A nucleophilic addition (AN) reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile.
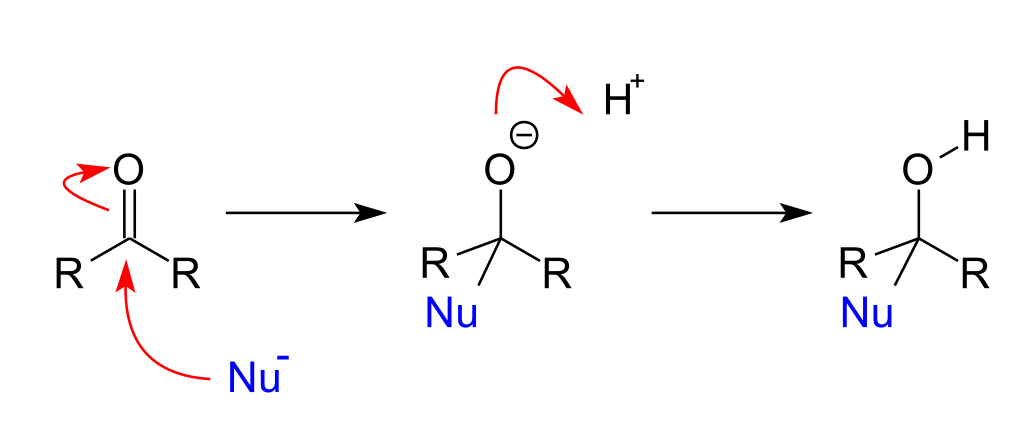
Nucleophilic addition reactions of nucleophiles with electrophilic double or triple pi bonds create a new carbon centre with two additional single or sigma bonds.
Mechanism
A stepwise ionic addition reaction involves nucleophilic attack at carbon as a first step, often catalysed by bases, which generate the required nucleophile. For example, consider the addition of some weakly acidic reagent \[\ce{H-X}\] to an alkene. In the presence of a strong base (\[\ce{OH-}\]), \[\ce{H-X}\] could give up its proton to form the conjugate base \[\ce{X-}\], which is expected to be a much better nucleophile than \[\ce{HX}\], \[\ce{H-X + OH- <=> H2O + X-}\].
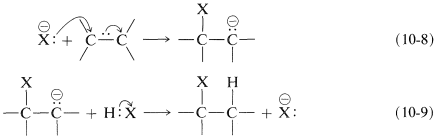
What can follow with an alkene is an ionic chain reaction with the following two propagating steps. First, the nucleophile (\[\ce{X-}\]) attacks at carbon to form a carbanion intermediate. Second, electrophilic transfer of a proton from \[\ce{H-X}\] to the carbanion forms the adduct and regenerates the nucleophile. The overall reaction is the addition of \[\ce{H-X}\] to the double bond.
Reaction of aldehydes and ketones with \[\ce{HCN}\]
Hydrogen cyanide adds across the carbon-oxygen double bond in aldehydes and ketones to produce compounds known as hydroxynitriles.
For instance, with ethanal (an aldehyde) you get 2-hydroxypropanenitrile:
or with propanone (a ketone) you get 2-hydroxy-2-methylpropanenitrile:
Similar to what we've described above, the \[\ce{C=O}\] double bond is highly polar, and the slightly positive carbon atom is attacked by the cyanide ion acting as a nucleophile:
The negative ion formed then picks up a hydrogen ion from somewhere, for example, from a hydrogen cyanide molecule, water or even the \[\ce{H3O+}\] ions present in the slightly acidic solution.
For ethanal, the process is pretty much the same,
, then
.
Referenced by:
No backlinks found.