carbocation
Carbocation
Carbocation is a general term for ions with a positively charged carbon atom. In the present-day definition given by the IUPAC, a carbocation is any even-electron cation (cation with fully paired outer-shells) with significant partial positive charge on a carbon atom.
Structure
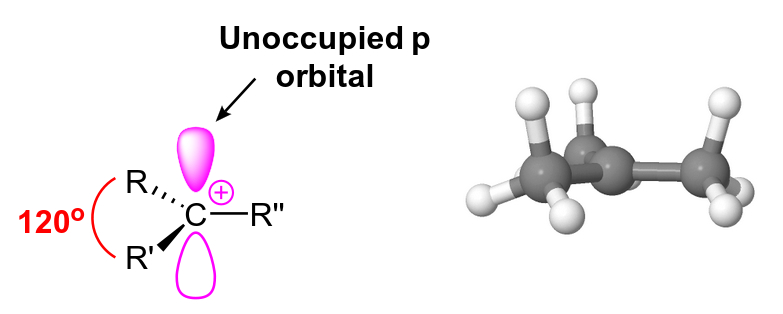
Carbocations typically have three substituents which makes the carbon sp2-hybridised and given the overall molecular a trigonal planar geometry. The carbocation's substituents are all in the same plane and have a bond angle of 120 degrees between them. The carbon atom in the carbocation is electron deficient as it only has six valence electrons which are used to form three sigma covalent bonds with the substituents. The carbocation carbon has an unoccupied p orbital which is perpendicular to the plane created by the substituents. The p orbital can easily accept electron pairs during reactions making carbocations excellent.
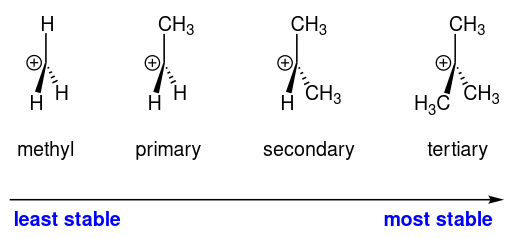
Carbocations have designations based on the number of alkyl groups attached to the carbocation carbon. Three alkyl groups attached is called a tertiary \[3^{\circ}\] carbocation, two is called a secondary \[2^{\circ}\] carbocation, one is called a primary \[1^{\circ}\] carbocation. If there are no alkyl groups attached, then it's a methyl carbocation.
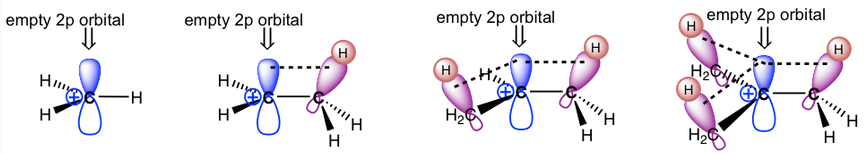
This can be explained simply with hyperconjugation (see below).
Stability
By being a reactive intermediate of the electrophilic addition mechanism, the stability of a carbocation has a direct effect on the reaction.

A positively charged species such as a carbocation is very electron-poor, and thus anything which donates electron density will help to stabilise it. Conversely, a carbocation will be stabilised by an electron withdrawing group.
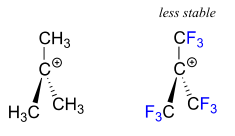
Common electron withdrawing groups are fluorine atoms and carbonyl groups (due to the polarity of the \[\ce{C=O}\] double bond).
Experimental evidence has shown that a carbocation becomes more stable as the number of alkyl (\[\ce{-CH3}\]) substituents increases.
Alkyl groups stabilizes carbocations for two main reasons. The first is that the electrons in covalent bonds are shifted towards an nearby atom with a higher electronegativity. In this case, the positively charged carbocation draws in electron density from the surrounding substituents thereby gaining stabilization by slightly reducing its positive charge. Alkyl groups are more effective at inductively donating electron density than a hydrogen because they are larger, more polarizable, and contain more bonding electrons. As more alkyl groups are attached to the carbocation, more inductive electron donation occurs and the carbocation becomes more stable.
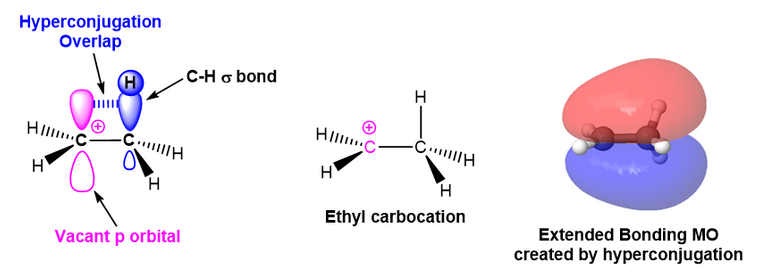
The second reason is through hyperconjugation. Hyperconjugation is an electron donation that occurs from the parallel overlap of p-orbitals with adjacent hybridized orbitals participating in sigma bonds. The idea here is that the electrons in the \[\ce{C-H}\] sigma bonds can overlap with the empty p-orbital of the carbocation as they are coplanar. This electron donation serves to stabilize the carbocation. As the number of alkyl substituents increases, the number of sigma bonds available for hyperconjugation increases, and the carbocation tends to become more stabilized.
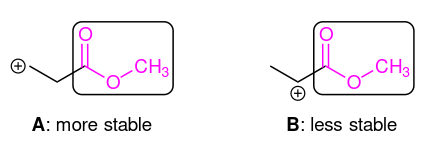
However, it is not always that a high substitution corresponds to more stability. If electron-withdrawing groups near to the carbocation, it can also acts destabilisers. As you can see above, species B is less stable as it's closer to the carbonyl group. Naturally, the strength of the effect decreases rapidly as the number of intermediary (covalent) bonds increases, thus species A is comparatively more stable.
Resonance
Stabilization of a carbocation can also occur through resonance effects, and as a rule are more powerful than effects of EDGs.
Consider the simple case of a benzylic carbocation.
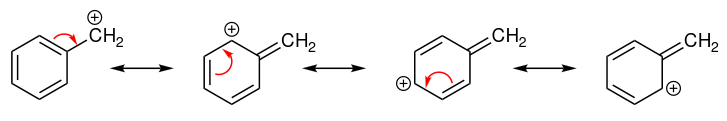
This carbocation is comparatively stable. In this case, electron donation is a resonance effect. Three additional resonance structures can be drawn for this carbocation in which the positive charge is located on one of three aromatic carbons. The positive charge is thus not isolated on the benzylic carbon, rather it is delocalized around the aromatic structure. This delocalisation of charge results in significant stabilization, making it significantly more stable than tertiary alkyl carbocations.
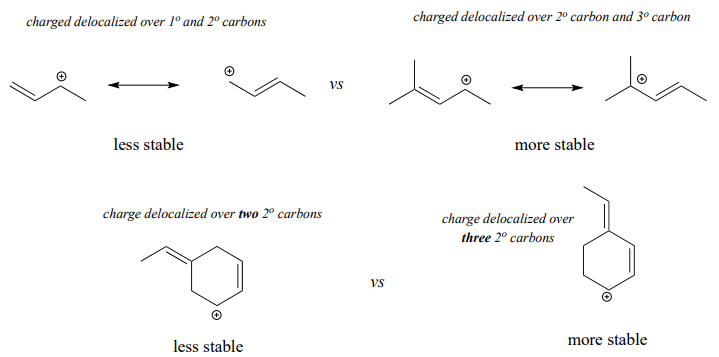
Predicting relative carbocation stability