molecular dipole
Molecular dipoles
Many molecules have bond dipole moments due to non-uniform distributions of positive and negative charges on the various atoms. Such is the case with polar compounds like hydrogen fluoride (\[\ce{HF}\]), where electron density is shared unequally between atoms. Therefore, a molecule's dipole is an electric dipole with an inherent electric field that should not be confused with a magnetic dipole, which generates a magnetic field.
Permanent dipoles
Occurs when two atoms in a molecule have substantially different electronegativity: One atom attracts electrons more than another, becoming more negative, while the other atom becomes more positive. A molecule with a permanent dipole moment is also known as a polar molecule.
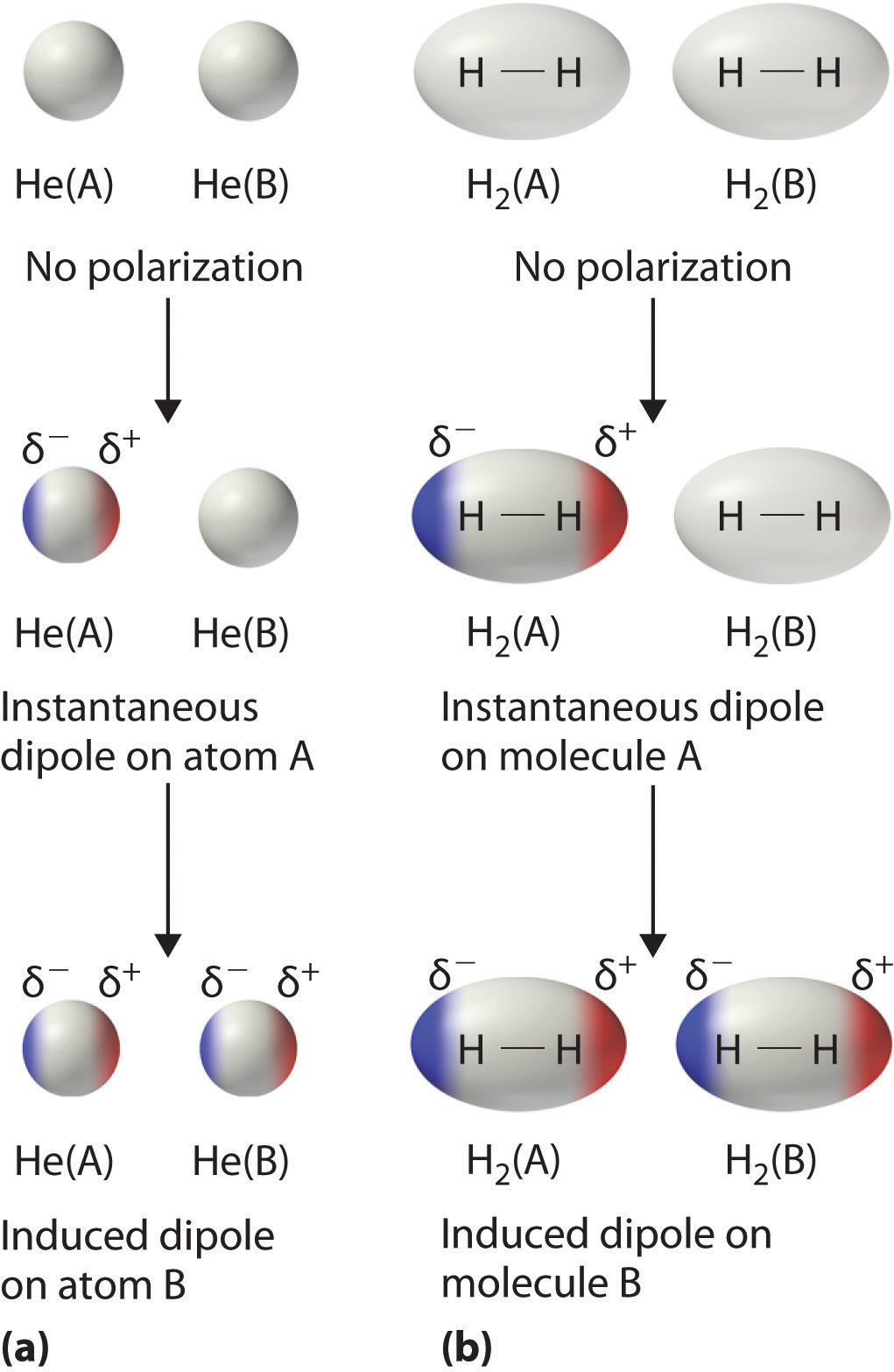
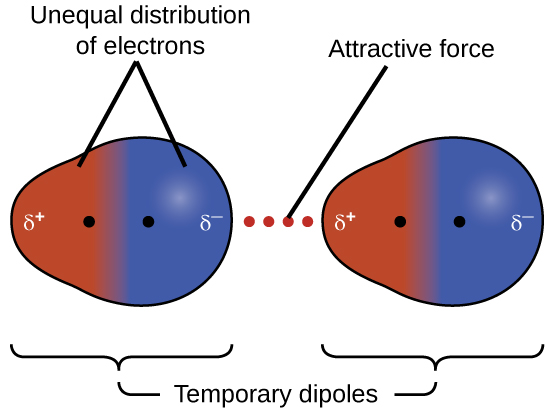
Instantaneous dipoles
Occurs due to the possibility that at any given time, electrons may happen to be more concentrated in one place than another in a molecule, creating a temporary dipole. These dipoles are smaller in magnitude than permanent dipoles.
Induced dipoles
Can occur when one molecule with a permanent or instantaneous dipole repels another molecule's electrons, inducing a dipole moment in that molecule.