cis-trans isomerism
Cis-trans isomerism
Known also as geometric isomerism, describes certain arrangements of atoms within molecules. Cis-trans isomers are stereoisomers, i.e. pairs of molecules which have the same formula but whose functional groups are in different orientations in three dimensional space.
Cis indicates that the functional groups are on the same side of some plane, while trans conveys that they are on opposing sides.
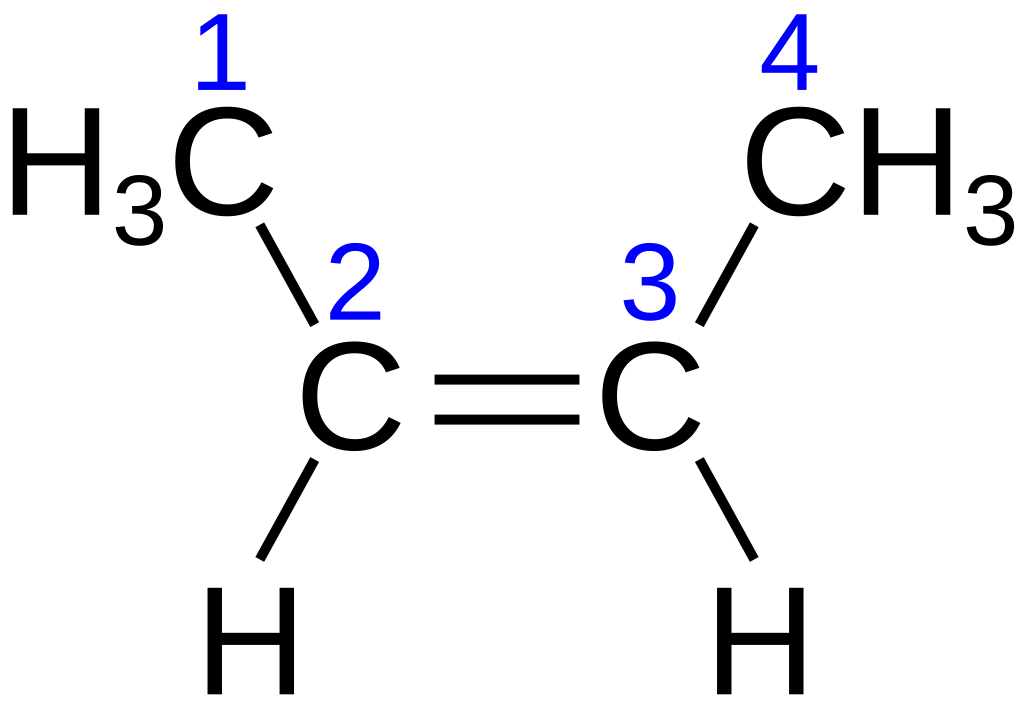
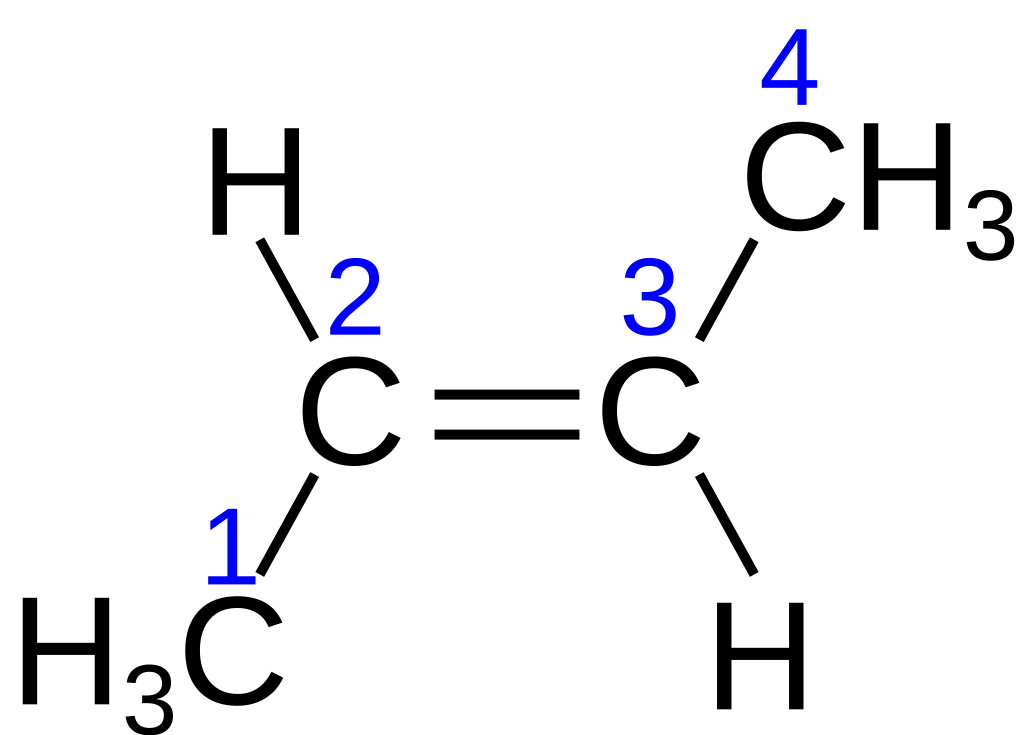
The first image shows cis-but-2-ene (same side of the plane) while the second image shows trans-but-2-ene (opposing sides).
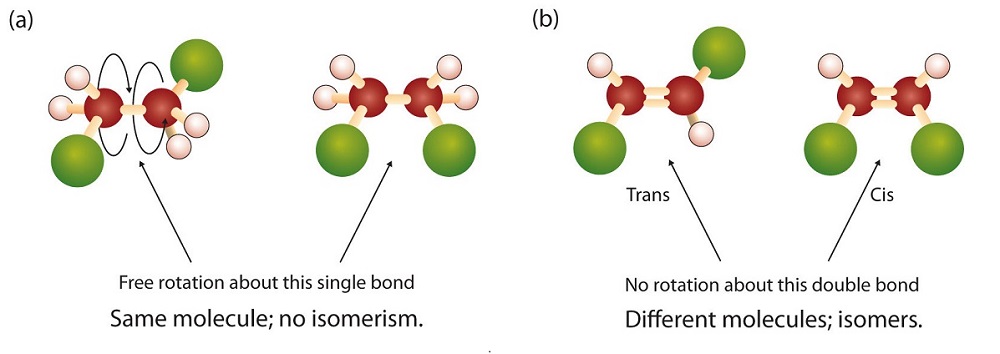
Though, one must be careful when it comes to single \[\ce{C-C}\] bonds in alkanes. For instance, in 1,2-dichloroethane, there is free rotation about the \[\ce{C-C}\] bond, thus no matter how you twist it they represent exact the same molecule, which makes them not isomers. This is contrasted from 1,2-dichloroethene, where the \[\ce{C=C}\] double is rigid, making rotation impossible without rupturing the double bond.
Cyclic compounds
If you have a ring of carbon atoms there will also be no possibility of rotation about any of the carbon-carbon bonds. Cyclohexane is a simple example:
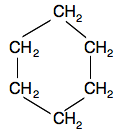
Now suppose you replaced two of the hydrogen in the cyclohexane molecule by two bromine atoms:
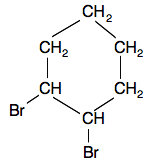
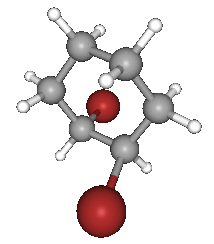
As you can see, the shape around each carbon atom connected to the bromine atoms are now tetrahedral, which means it is possible for them to have cis-trans isomers.
Effects of cis-trans isomerism on physical properties
Generally, the trans isomer has a higher melting point but a lower boiling point, while the cis isomer will have a lower melting point but a higher boiling point.
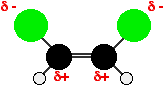
This can be explained with the fact that boiling point increases, i.e. in the cis isomer, one side of the molecule will have a slight negative charge while the other is slightly positive. The molecule is therefore polar. Because of the polarity, there will be dipole-dipole interactions as well as London dispersion forces which needs extra energy to break. That will raise the boiling point.
Now, as to why trans isomers have a higher melting point when logically cis isomers should be the one having a higher melting point. In order for the intermolecular forces to work well in the solid, the molecules must be able to pack together efficiently. Trans isomers pack better than cis isomers as the "U" shape of the cis isomer does not pack as well as the straighter shape of the trans isomer. The poorer packing in the cis isomers means that the intermolecular forces are not as effective as they should be so less energy is needed to melt the molecule, making the melting point lower.
Referenced by:
No backlinks found.