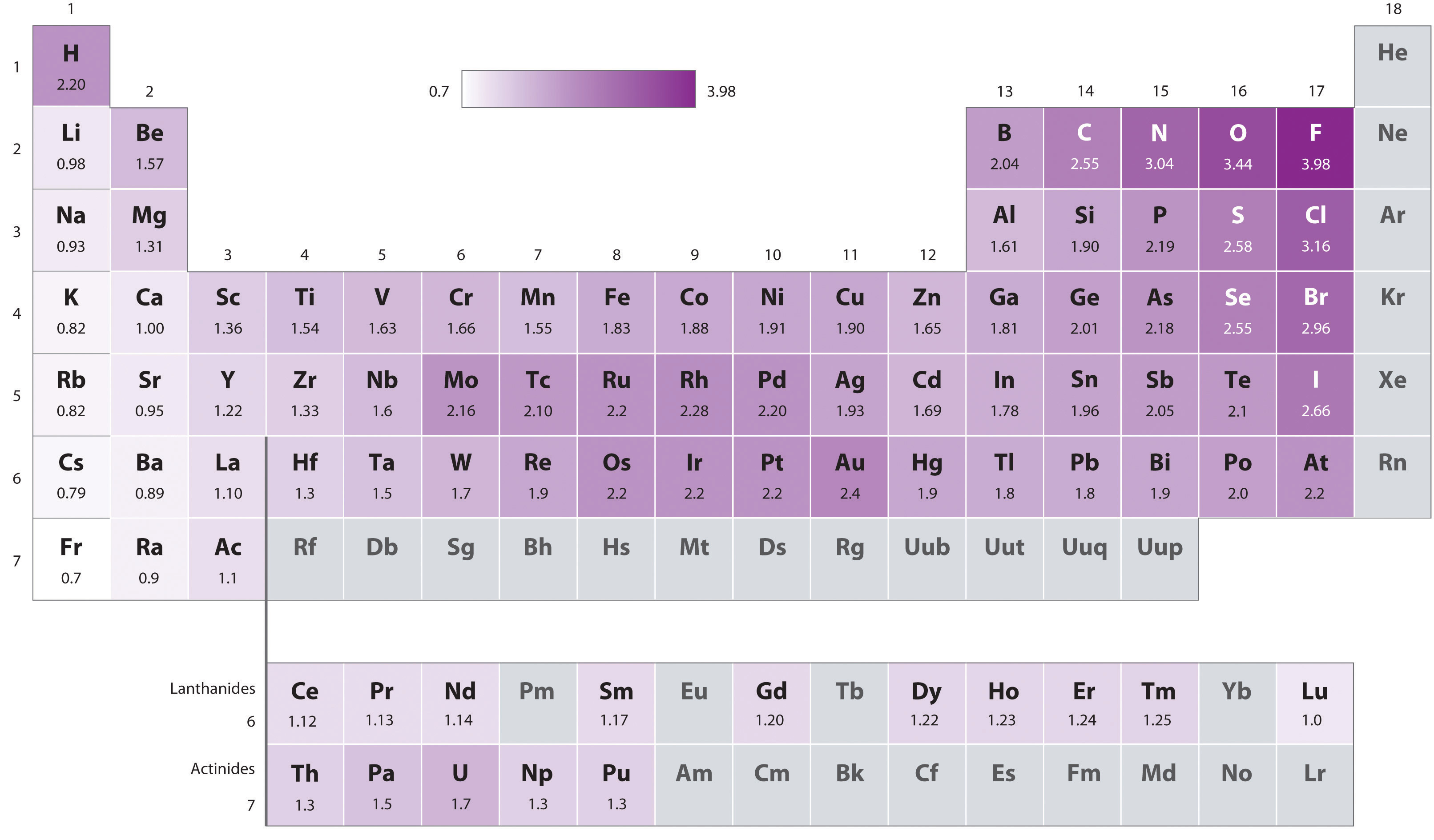
electronegativity
Electronegativity
Electronegativity of an atom is a relative value of that atom's ability to attract election density toward itself when it bonds to another atom. The higher the electronegative of an element, the more that atom will attempt to pull electrons towards itself and away from any atom it bonds to.
The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to cesium and francium which are the least electronegative at 0.7.
The attraction that a bonding pair of electrons feels for a particular nucleus depends on:
- Nuclear charge
- Distance of electron from the nucleus
- Electron shielding by inner subshells (effective nuclear charge)
Trends
Across the period
The positively charged protons in the nucleus attract the negatively charged electrons. As the number of protons in the nucleus increases, the electronegativity or attraction will increase. Therefore electronegativity increases from left to right in a row in the periodic table. This effect only holds true for a row in the periodic table because the attraction between charges falls off rapidly with distance.
Electronegativity increases across a period due to the increasing number of protons in the nucleus. Comparing sodium and chlorine demonstrates this trend. Both elements have their bonding electrons in the \[n\] = 3-level, shielded from the nucleus by the inner 1s, 2s, and 2p electrons. However, chlorine's nucleus contains six more protons than sodium's. This substantial increase in nuclear charge means that chlorine exerts a much stronger attraction on the bonding electrons.
Across the group
As you go down a group, electronegativity decreases because the bonding pair of electrons is increasingly distant from the attraction of the nucleus. Consider the hydrogen fluoride and hydrogen chloride molecules:
The bonding pair is shielded from the fluorine's nucleus only by the 1s2 electrons. In the chlorine case it is shielded by all the 1s22s22p6 electrons. In each case there is a net pull from the center of the fluorine or chlorine of +7. But fluorine has the bonding pair in the 2-level rather than the 3-level as it is in chlorine. If it is closer to the nucleus, the attraction is greater.
Predicting bonds with electronegativity
This section should be prefaced with the fact that there is not a single bond that is a hundred percent ionic or covalent. Though people have achieved very close to a hundred percent ionic bonds with complicated ingenuity, the point still stands.
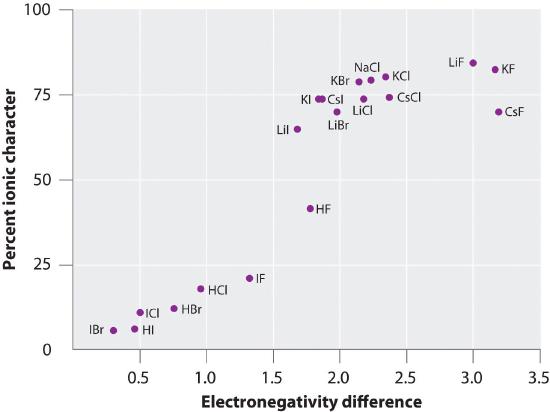
Here's a table that plots the percent ionic character of a bond as determined from measured bond dipole moment against the difference in electronegativity of the bonded atoms. In gas phase, even \[\ce{CsF}\], which has the largest possible difference in electronegativity between atoms is not 100 percent ionic.
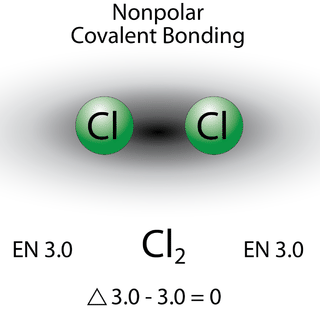
Nonpolar covalent bonds
A nonpolar covalent bond is a covalent bond in which the bonding electrons are shared equally (sometimes almost) between the two atoms. In a nonpolar covalent bond, the distribution of electrical charge is balanced between the two atoms. Molecules in which the electronegativity difference is very small (<0.4) are considered nonpolar covalent ( \[\Delta\text{EN}=3.0-2.8=0.2\] ).
Polar covalent bonds
A bond in which the electronegativity difference between the atoms is between 0.4 and 2.0 is called a polar covalent bond. A polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal. In a polar covalent bond, sometimes simply called a polar bond, the distribution of electrons around the molecule is no longer symmetrical.
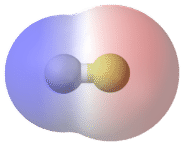
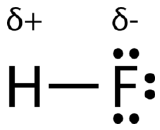
In the polar covalent bond of HF, the electron density is unevenly distributed. There is a higher density (red) near the fluorine atom, and a lower density (blue) near the hydrogen atom.
Ionic bonds
Anything with a electronegativity difference of >2.0 is most likely an ionic bond.