London dispersion forces
London dispersion forces
London dispersion forces (also known as dispersion forces, London forces, instantaneous dipole-induced dipole forces), are a type of intermolecular force acting between atoms and molecules that are normally electrically symmetric; that is, the electrons are symmetrically distributed with respect to the nucleus. The effects of London dispersion forces are most obvious in systems that are nonpolar (do note London forces also occur in polar molecules), such as hydrocarbons and highly symmetric molecules like bromine or iodine.
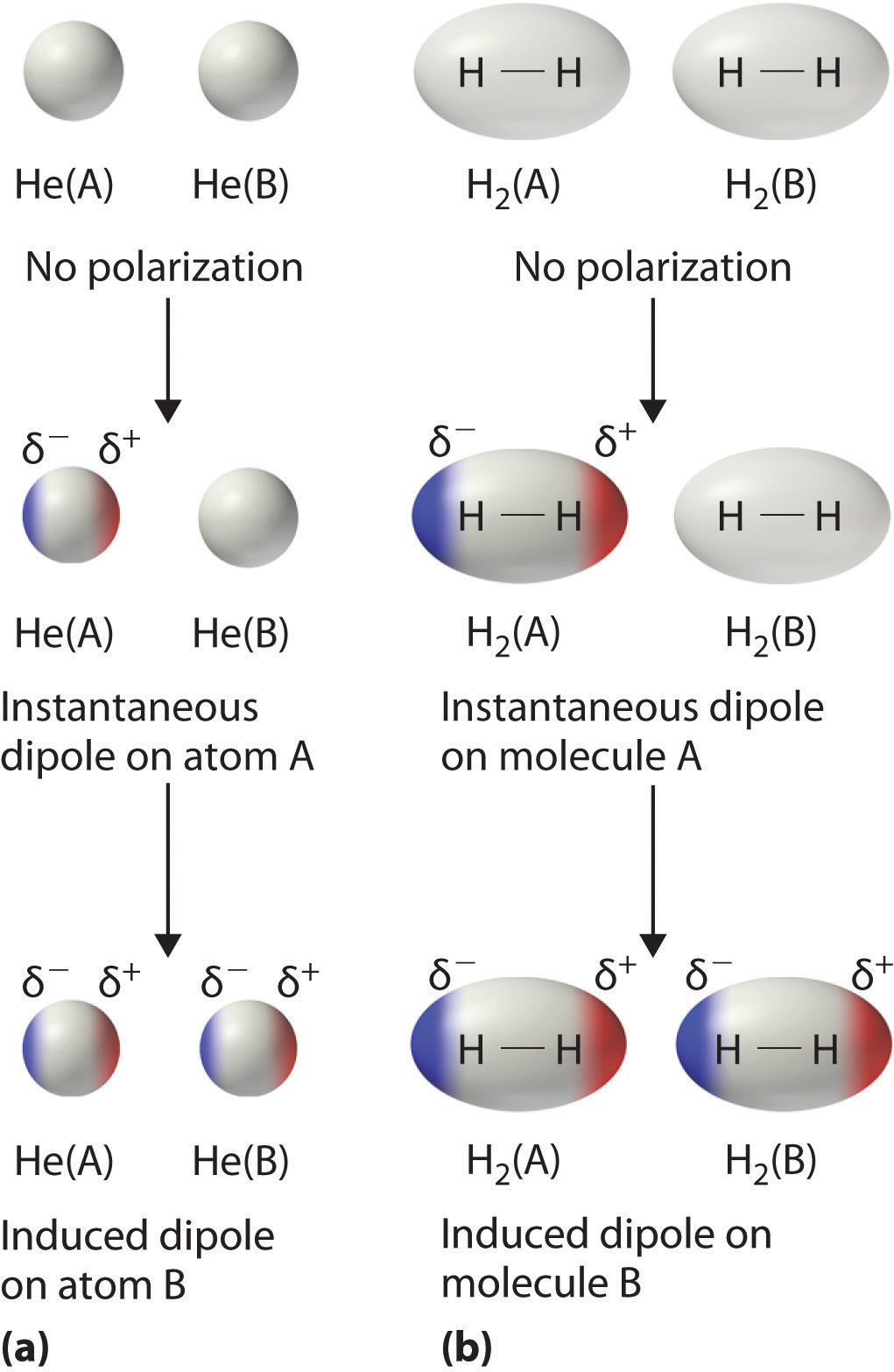
The electron distribution around an atom or molecule undergoes fluctuations in time. These fluctuations create instantaneous electric fields which are felt by other nearby atoms and molecules, which in turn adjust the spatial distribution of their own electrons. An atom with a large number of electrons will have a greater associated London force than an atom with fewer electrons. The net effect is that the fluctuations in electron positions in one atom induce a corresponding redistribution of electrons in other atoms, such that the electron motions become correlated.
In more simple terms, the chance that an electron of an atom is in a certain area in the electron cloud at a specific time is called the "electron charge density." Since there is no way of knowing exactly where the electron is located and since they do not all stay in the same area 100 percent of the time, if the electrons all go to the same area at once, a dipole is formed momentarily. Even if a molecule is nonpolar, this displacement of electrons causes a nonpolar molecule to become polar for a moment, this is called an instantaneous dipole.
The strengths of London dispersion forces also depend significantly on molecular shape because shape determines how much of one molecule can interact with its neighboring molecules at any given time. For example, 2,2-dimethylpropane and n-pentane, both of which have the empirical formula \[\ce{C5H12}\]. 2,2-dimethylpropane is almost spherical, with a small surface area for intermolecular interactions, whereas n-pentane has an extended conformation that enables it to come into close contact with other n-pentane molecules. As a result, the boiling point of 2,2-dimethylpropane (9.5 C) is more than 25 C lower than the boiling point of n-pentane (36.1 C). (See image below)
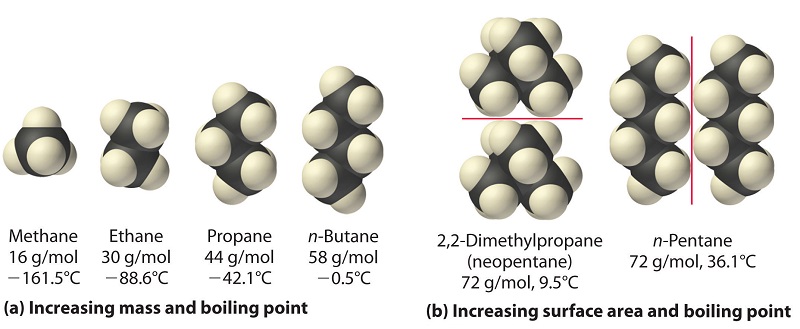
Evidence of sass and surface area affecting the strength of London dispersion forces.
(a) In this series of four simple alkanes, larger molecules have stronger London forces between them than smaller molecules do, and consequently have higher boiling points.
(b) Linear n-pentane molecules have a larger surface area and stronger intermolecular forces than spherical 2,2-dimethylpropane molecules. As a result, 2,2-dimethylpropane is a gas at room temperature, whereas n-pentane is a volatile liquid.