hydrogen bond
Hydrogen bond
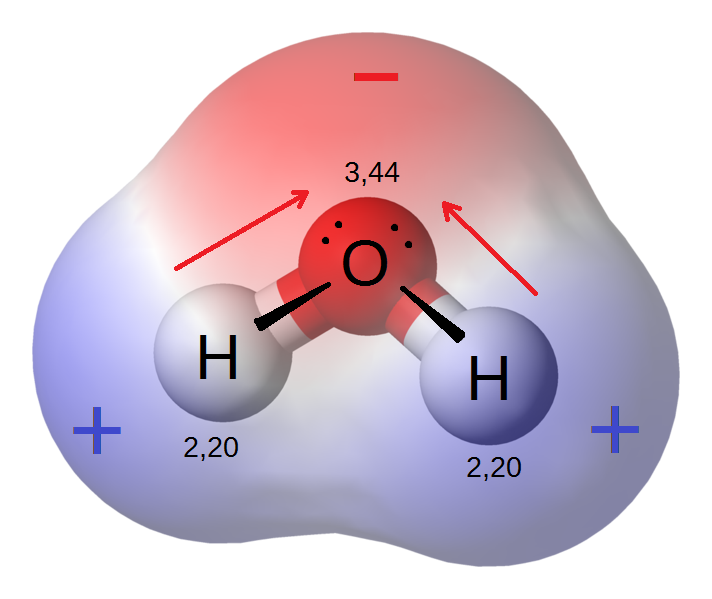
A hydrogen bond, or H-bond, is primarily an electrostatic force of attraction between a hydrogen atom which is covalently bonded to a more electronegative atom, called the proton donor, and another electronegative atom bearing a lone pair of electrons, called the proton acceptor. The most frequent donor and acceptor atoms are the period 2 elements nitrogen, oxygen, and fluorine. Hydrogen bonds can be intermolecular (occurring between separate molecules) or intramolecular (occurring among parts of the same molecule). It is somewhat stronger than a van der Waals interaction, but weaker than fully covalent or ionic bonds. Usually, any molecule which has a hydrogen atom attached directly to an oxygen or a nitrogen is capable of hydrogen bonding.
When hydrogen is attached directly to a highly electronegative atom, the molecule becomes polar. However, the proton donor can not completely remove the valence electron from hydrogen and form an ion because there are no core electrons in hydrogen. Removing the hydrogen's 1s electron would produce a proton, whose small size results in a high charge density that would pull back the electron. The electronegative atom's pull on the valence electron "deshields" the hydrogen's proton resulting in a \[\delta+\] charge over a small area. The highly electronegative atom now has a \[\delta-\] charge and if it has a lone pair of electrons, it will be attracted to the "deshielded" proton of another hydrogen and create a hydrogen bond. It should also be noted that the small size of the hydrogen allows both molecules to bind closely, resulting is a strong bonding interaction.
It is to note that despite it being called a "bond", it is in actuality a type of attraction that is approximately 90% electrostatic and 10% covalent. The covalency is due to an observed 1% of an electron transferred from the lone pair into the \[\ce{O-H}\] donor orbital (see Isaacs et al. (2000)).
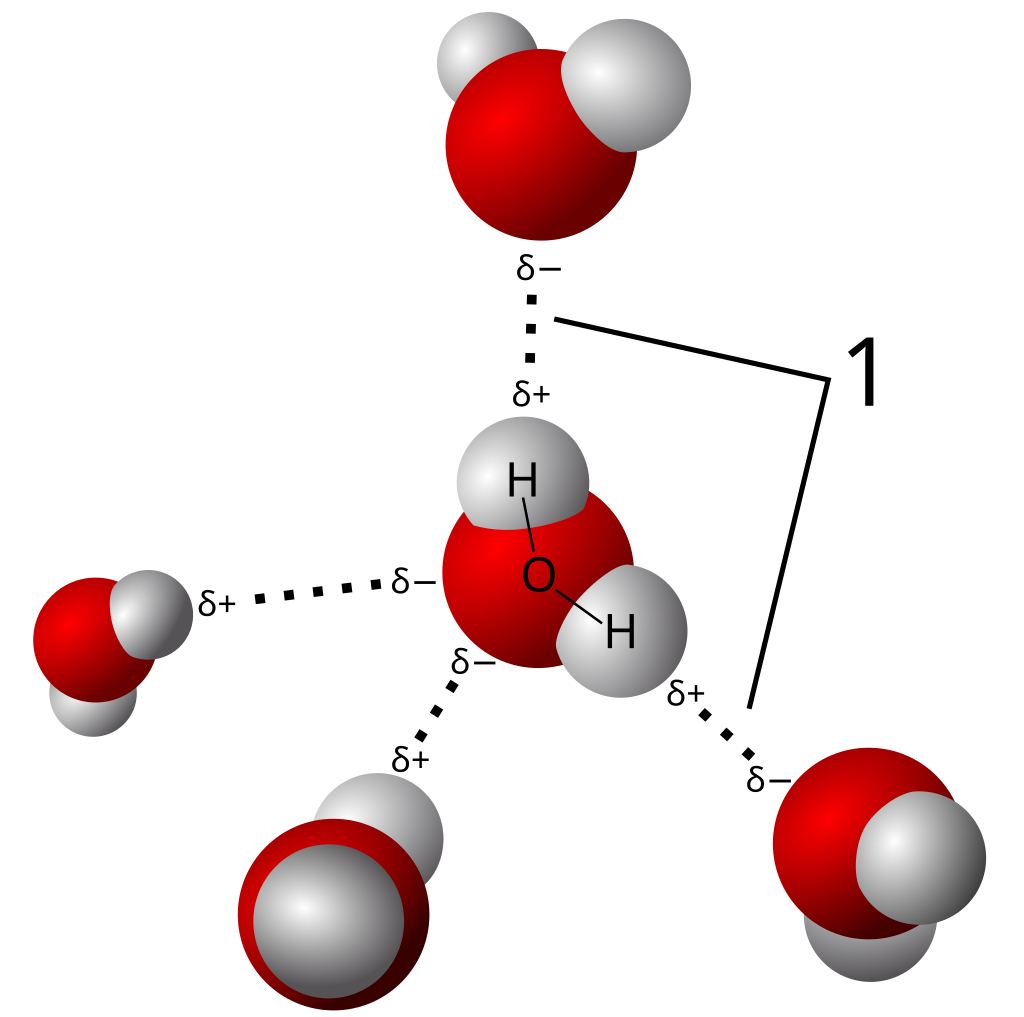
Hydration of negative ions (Red indicates a lone pair):
Factors preventing hydrogen bonds
The simplified explanation is that hydrogen bonding cannot occur without significant electronegativity differences between hydrogen and the atom it is bonded to. Thus, we see molecules such as \[\ce{PH3}\], which do not participate in hydrogen bonding. \[\ce{PH3}\] exhibits a trigonal pyramidal molecular geometry like that of ammonia, but unlike \[\ce{NH3}\] it cannot hydrogen bond. This is due to the similarity in the electronegativities of phosphorous and hydrogen. Both atoms have an electronegativity of 2.1, and thus, there is no dipole moment. This prevents the hydrogen atom from acquiring the partial positive charge needed to hydrogen bond with the lone electron pair in another molecule.
The size of donors and acceptors can also affect the ability to hydrogen bond. This can account for the relatively low ability of \[\ce{Cl}\] to form hydrogen bonds. When the radii of two atoms differ greatly or are large, their nuclei cannot achieve close proximity when they interact, resulting in a weak interaction.
The more detailed explanation as to why you usually only see hydrogen bonding in smaller atoms and not larger atoms like chlorine (the hydrogen bonding in actuality still exists, just very weak, see Goutev & Matsuura (2001)) is that taking the case of chlorine, the lone pairs of chlorine are at 3-level, which is too high. High here refers to the high energy of the 3p orbital of chlorine, or in a more literal sense being a bit farther from the nucleus, thus the 3p cloud overlaps poorly with the tight 1s cloud of the hydrogen. Since the hydrogen bond is a mostly dipole-dipole attraction and because the force between two dipoles scales roughly \[\frac{1}{r^{4}}\], a larger atom implies a significantly weaker dipole-dipole attraction.
Evidence
Many elements form compounds with hydrogen. If you plot the boiling points of the compounds of the group 14 elements with hydrogen, you find that the boiling points increase as you go down the group (as they are nonpolar, therefore does not form hydrogen bonds).
The increase in boiling point happens because the molecules are getting larger with more electrons, and so van der Waals dispersion forces become greater. However, if you repeat this exercise with the compounds of the elements in groups 15, 16, and 17 with hydrogen, something odd happens.
Although the same reasoning applies for group 4 of the periodic table, the boiling point of the compound of hydrogen with the first element in each group is abnormally high. Therefore there must be some additional intermolecular forces of attraction, requiring significantly more heat energy to break the IMFs. These relatively powerful intermolecular forces are the evidence of the existence of hydrogen bonds.
Hydrogen bonding in alcohols
Ethanol, \[\ce{CH3CH2-O-H}\], and methoxymethane, \[\ce{CH3-O-CH3}\], both have the same molecular formula, \[\ce{C2H6O}\]. The van der Waals attractions are similar, however ethanol has a hydrogen atom attached directly to an oxygen; here the oxygen still has two lone pairs like a water molecule. Hydrogen bonding can occur between ethanol molecules, although not as effectively as in water. The hydrogen bonding is limited by the fact that there is only one hydrogen in each ethanol molecule with sufficient \[\delta+\] charge. In methoxymethane, the lone pairs on the oxygen are still there, but the hydrogens are not sufficiently \[\delta+\] for hydrogen bonds to form.
Guide to hydrogen bond strength
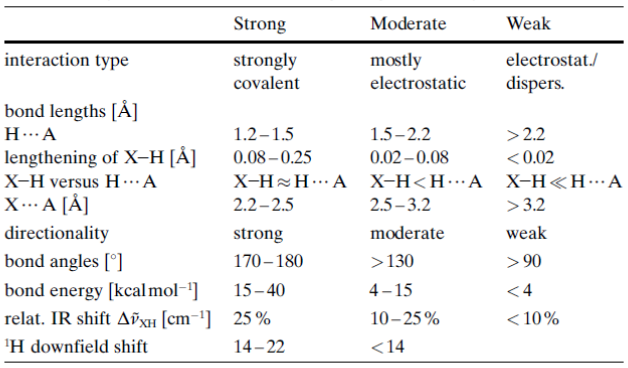
(Dannenberg, J. J., 1998)
- \[\ce{H . . . A}\] is the distance from hydrogen to the acceptor atom \[\ce{A}\], a shorter \[\ce{H . . . A}\] bond length implies stronger Couloumb attraction and more orbital overlap.
- Lengthening of \[\ce{X-H}\] shows how much the donor bond \[\ce{X-H}\] stretches when the hydrogen bond forms.
- \[\ce{X-H}\] versus \[\ce{H . . . A}\] tells us roughly the relative distance of the proton in between both heavier atoms.
- \[\ce{X . . . A}\] tells us the separation distance between the two heavy atoms.
- Since the hydrogen bonds favour linearity, distortion weakens overlap and electrostatics, thus strong hydrogen bonds are at an almost straight angle while weak ones are bent.
- The bond energy is the approximate enthalpy of a single hydrogen bond.