chemical properties of group 2 elements
Chemical properties of group 2 elements
Reactions with oxygen
Group 2 elements react with oxygen to form metal oxides. The general reaction is given as: \[\ce{2 X + O2 -> 2 XO}\]. The reaction is identical for all group 2 elements.
\[\ce{2 Ca + O2 -> 2 CaO}\]
Half-equations: \[\ce{Ca -> Ca^{2+} + 2 e^{-}}\] (oxidation), \[\ce{O2 + 4 e^{-} -> 2 O^{2-}}\] (reduction)
Reactions with water
Group 2 elements reduce water into hydrogen gas. The general equation is given as: \[\ce{X(s) + 2 H2O(l) -> X(OH)2(s) + H2(g)}\].
\[\ce{Sr + 2 H2O -> Sr(OH)2 + H2}\]
Half-equations: \[\ce{Sr -> Sr^{2+} + 2 e^{-}}\](oxidation), \[\ce{2 H2O + 2 e^{-} -> 2 OH^{-} + H2}\]
Magnesium: Magnesium reacts very slowly in while cold water, \[\ce{Mg + 2 H2O -> Mg(OH)2 + H2}\], but reacts quicker when steam is passed over heated magnesium, \[\ce{Mg + H2O -> MgO + H2}\]
As the reactivity increased down group 2, starting with calcium, it is capable of reacting quickly in cold water
Reactions with dilute acids
With dilute hydrochloric acids: \[\ce{X(s) + 2 HCl(aq) -> XCl2(aq) + H2(g)}\]
With dilute sulfuric acid: \[\ce{X(s) + H2SO4(aq) -> XSO4(aq) + H2(g)}\]
Reactions of oxides, hydroxides and carbonates with water
Group 2 oxides are soluble with water, with the exception of magnesium oxide, which is only sparingly soluble. \[\ce{XO(s) + H2O(l) -> X(OH)2(aq)}\]
Oxides: Solubility increases down the group with increasing pH (increasingly alkali). (Due to increasing atomic radii, thus decreasing lattice energy)
Hydroxides: Solubility increases down the group, \[\ce{X(OH)2(s) -> X^{2+}(aq) + 2 OH- (aq)}\]. (Explanation is similar to oxides)
Solubility and pH of group 2 hydroxides:
| Hydroxide | pH |
|---|---|
| \[\ce{Mg(OH)2}\] | 8.0 |
| \[\ce{Ca(OH)2}\] | 12.0 |
| \[\ce{Sr(OH)2}\] | 13.0 |
| \[\ce{Ba(OH)2}\] | 13.5 |
Carbonates: Carbonates of group 2 elements are insoluble in water (High lattice energy and thermodynamically stable)
Reactions of oxides, hydroxides and carbonates with dilute acids
Group 2 oxides and hydroxides are basic (as when they are added to water they produce \[\ce{OH-}\] ions, making them a Lewis base) and neutralise acids.
For oxides: \[\ce{O^{2-} + 2 H+ -> H2O}\]
For hydroxides: \[\ce{OH + H+ -> H2O}\]
Oxides: \[\ce{MgO(s) + 2 HCl(aq) -> MgCl2(aq) + H2O(l)}\]
Hydroxides: \[\ce{Ca(OH)2(s) + H2SO4(aq) -> CaSO4(aq) + 2 H2O(l)}\]
Carbonates: \[\ce{MgCO3(s) + H2SO4(aq) -> MgSO4(aq) + H2O(l) + CO2(g)}\]
Note: Reactions with sulfuric acid and calcium oxide/hydroxide/carbonate is sparring soluble while starting from strontium it becomes insoluble in sulfuric acid as precipitates of insoluble group 2 sulfates form on the surface of the oxides/hydroxides/carbonates and prevent reactions from taking place
Solubility of hydroxides and sulfates
Hydroxides: Increasingly soluble as you go down the group
Sulfates: Becomes less soluble as you go down the group
Thermal decomposition of nitrates and carbonates
Nitrates: Decompose when heated to produce white metal oxide, brown nitrogen dioxide gas and oxygen gas, \[\ce{X(NO3)2(s) -> XO(s) + 4 NO2(g) + O2(g)}\].
Carbonates: Decompose when heated to produce metal oxide and carbon dioxide, \[\ce{XCO3(s) -> XO(s) + CO2(g)}\].
The thermal stability of the group 2 nitrates and carbonates increase down the group, meaning going down the group will require more and more energy to decompose it. This is because carbonate and nitrate ions have a resonant structure, which is a type of bonding in which combines of several contributing structures.
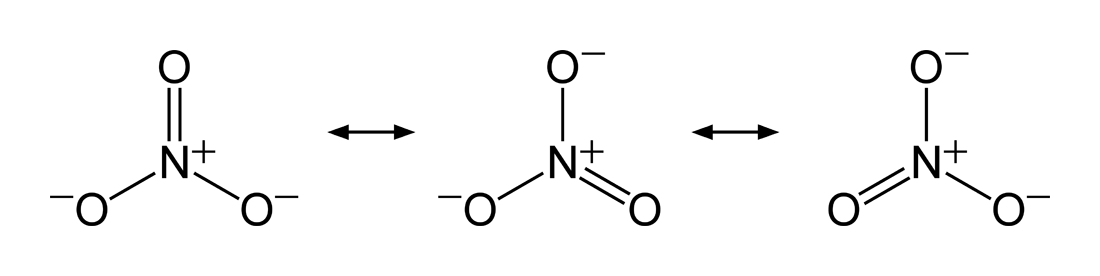
and 
Which results in:
 , where each oxygen has a \[-\frac{2}{3}\] charge and the nitrogen has +1 charge, (as the total charge of a nitratre ion is -1, which matches \[\left(3\cdot -\frac{2}{3}\right)+1 = -1\]),
, where each oxygen has a \[-\frac{2}{3}\] charge and the nitrogen has +1 charge, (as the total charge of a nitratre ion is -1, which matches \[\left(3\cdot -\frac{2}{3}\right)+1 = -1\]), 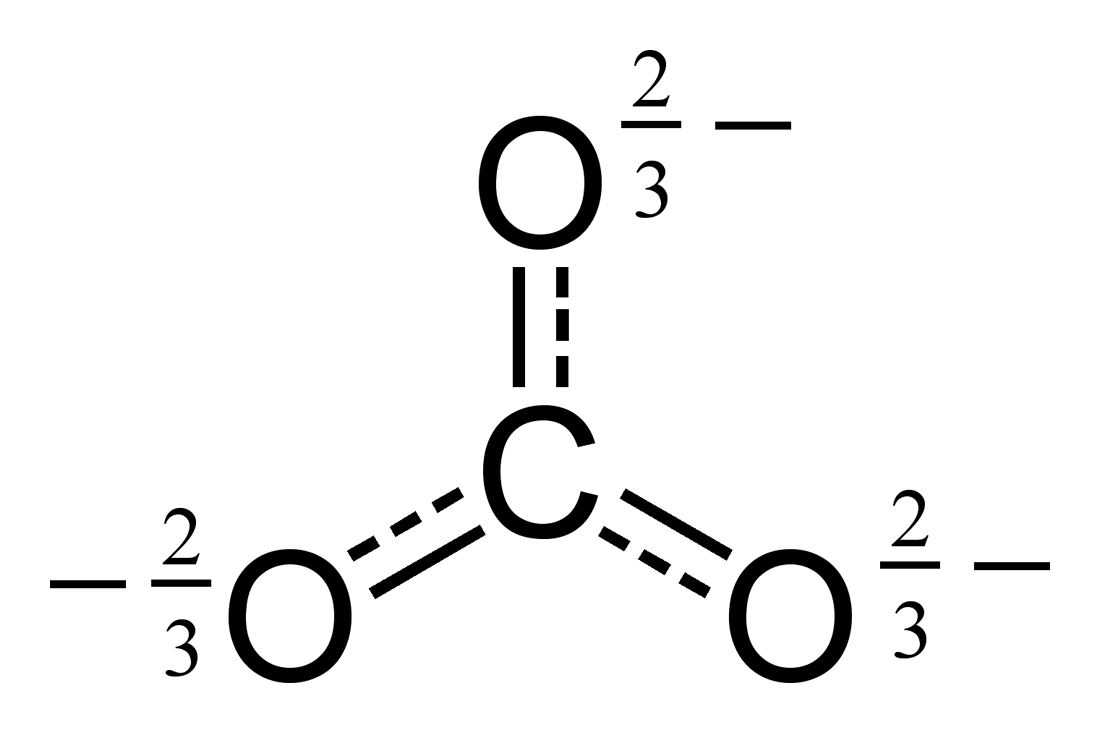 , where similar to nitrates, each oxygen has a \[-\frac{2}{3}\] charge, but since the carbon is neutral, carbonate ion has a -2 total charge.
, where similar to nitrates, each oxygen has a \[-\frac{2}{3}\] charge, but since the carbon is neutral, carbonate ion has a -2 total charge.
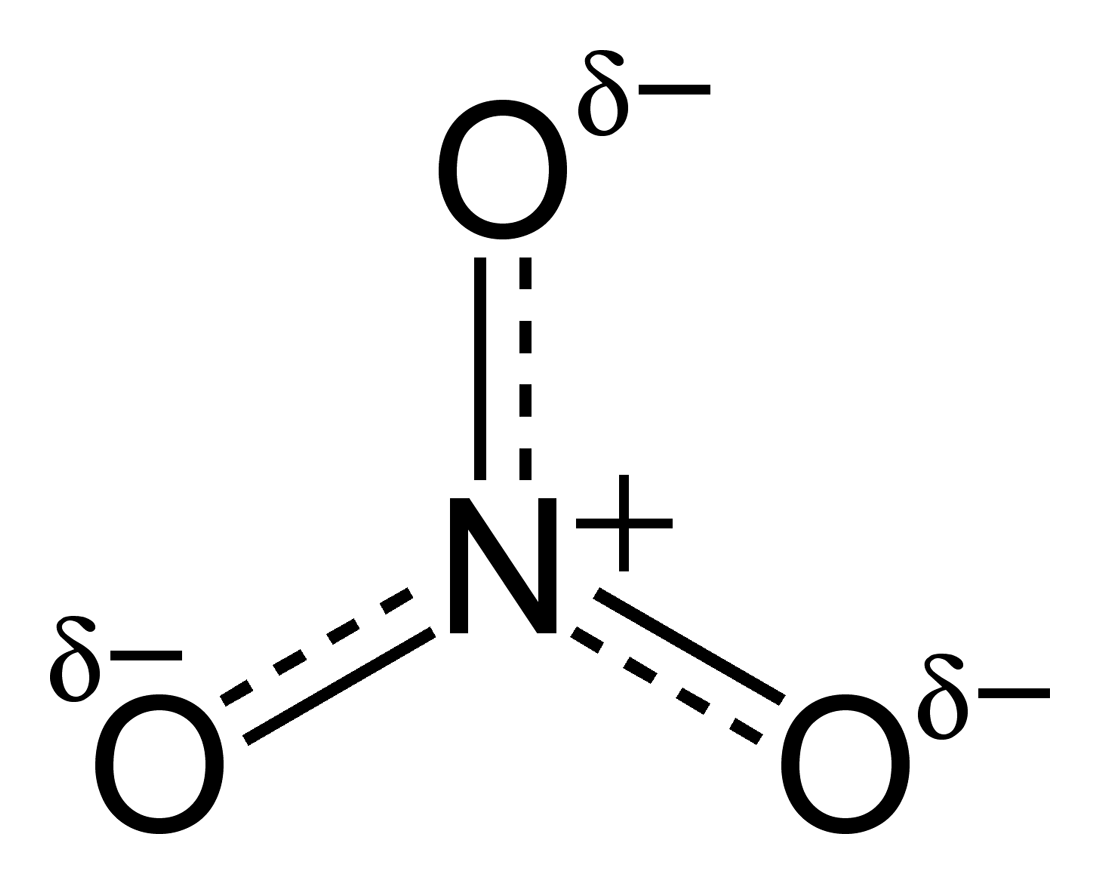
This causes the charges to be spread out (delocalised) across the structure and concentrating at the oxygen atoms:  . Now, when bonded to group 2 ions,
. Now, when bonded to group 2 ions,
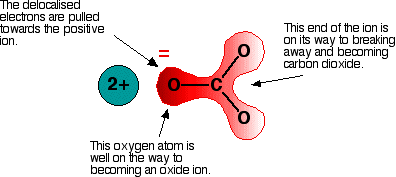 , which shows that the group 2 ion pulling the delocalised electrons towards it, turning the carbonate/nitrate ion polar. When this compound is heated, the \[\ce{O-C-O}\] or the \[\ce{O-N-O}\] will break free to form carbon dioxide/nitrogen dioxide and a group 2 metal oxide.
, which shows that the group 2 ion pulling the delocalised electrons towards it, turning the carbonate/nitrate ion polar. When this compound is heated, the \[\ce{O-C-O}\] or the \[\ce{O-N-O}\] will break free to form carbon dioxide/nitrogen dioxide and a group 2 metal oxide.
The smaller the positive ion is, the higher the charge density, and the greater effect it will have on the carbonate ion. As the positive ions get bigger as you go down group 2, they have less effect on the carbonate ions near them. To compensate for that, the heat required to persuade the carbon dioxide to break free and leave the metal oxide will increase, thus increasing thermal stability.
Referenced by:
No backlinks found.