free-radical addition
Free-radical addition
Free-radical addition is an addition reaction (a reaction in which two or more molecules combine to form a larger molecule) which involves free radicals. This reaction depend on one or more relatively weak bonds in a reagent. Under reaction conditions (typically heat or light), some weak bonds homolyse into radicals, which then induce further decomposition in their compatriots before recombination. Different mechanisms typically apply to reagents without such a weak bond.
Mechanism
The example used here would be the radical hydrobromination of an alkene.
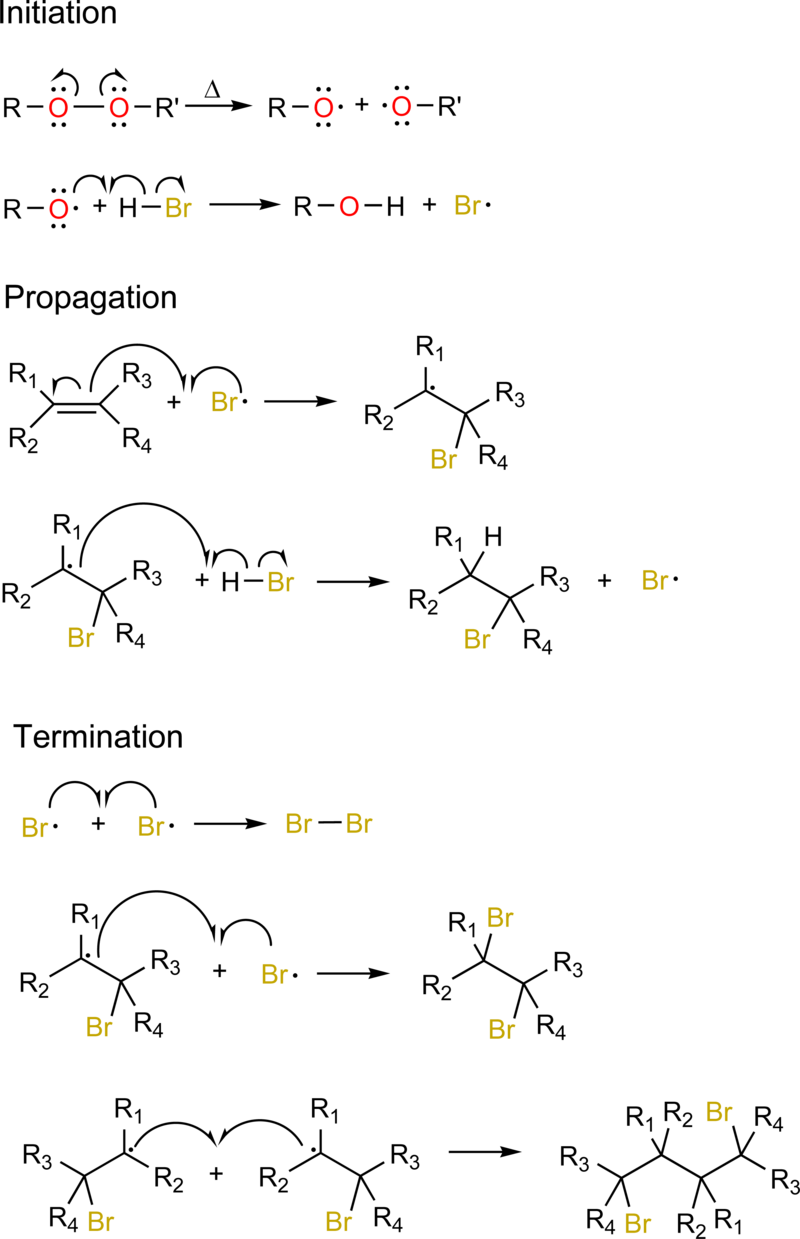
Initiation
This is the first step where a radical is created from a non-radical precursor (a stable molecule that can generate radicals when provided with sufficient energy). For example, Consider the decomposition of a peroxide (a molecule with an \[\ce{-O-O-}\] bond) under heat. The peroxide bond breaks apart to form two oxygen radicals: \[\ce{R-O-O-R ->[\text{Heat}] 2 R-O^{.}}\], and the \[\ce{O^{.}}\] is what makes it highly reactive.
Once the alkoxy radicals (\[\ce{RO^{.}}\]) are generated, they will initiate the radical chain reaction by interacting with hydrogen bromide \[\ce{HBr}\]: \[\ce{RO^{.} + HBr -> ROH + Br^{.}}\]. The reaction here is a homolytic cleavage, and it's energetically favorable as the bond energy for \[\ce{H-Br}\] is sufficiently low. The reason why the alkoxy radical attacks \[\ce{HBr}\] is due to it being highly reactive and constantly seeks to achieve a more stable electron configuration by abstracting a hydrogen atom from a suitable donor. HBr provides an ideal hydrogen donor due to the bond polarization, where hydrogen is \[\delta+\] and bromine is \[\delta-\].
Propagation
With bromine radicals \[\ce{Br^{.}}\] now present, the reaction enters the chain propagation phase, which involves two key steps: the addition of bromine radical to the alkene and the abstraction of a hydrogen atom by the bromoalkyl radical.
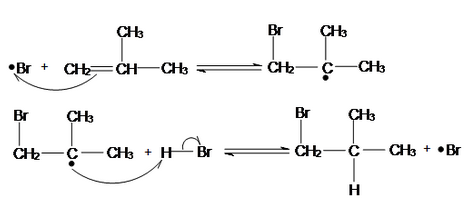
The bromine radical adds to the least substituted carbon of the alkene (in this case, 2-methylpropene \[\ce{CH2=CHCH3CH3}\]) resulting in a bromoalkyl radical, \[\ce{CH2Br-C^{.}CH3CH3}\]. This follows the anti-Markovnikov rule, where the bromine attaches to the least substituted carbon (the carbon with fewer alkyl groups), and the radical is formed on the more substituted carbon. This orientation is driven by the stability of the resulting radical.
The bromoalkyl radical then abstracts a hydrogen atom from another molecule of \[\ce{HBr}\] forming 1-bromo-2-methylpropane \[\ce{CH2BrCHCH3CH3}\] and regenerating a new bromine radical.
Termination
The reaction eventually reaches the chain termination phase, where two radicals combine to form a stable, non-radical product, effectively stopping the reaction cycle: \[\ce{Br^{.} + Br^{.} -> Br2}\]. Alternatively the bromine radical may combine with the carbon radical or two bromoalkyl radicals can combine to form a dimeric product.
Differentiating between electrophilic addition and radical addition
We can distinguish electrophilic and radical addition of \[\ce{H-Br}\] to alkenes by examining the regiochemistry of the addition product. While electrophilic addition has Markovnikov regiochemistry, free radical additon has the opposite regiochemistry that we will refer to as anti-Markovnikov addition. We show this contrasting regiochemistry in the electrophilic and radical additions of \[\ce{H-Br}\] to 1-methylcyclohexene:
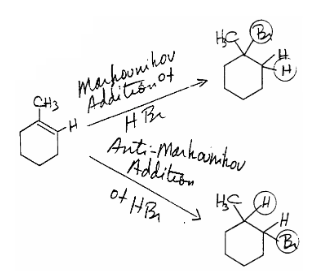
The difference in regiochemistry for \[\ce{H-Br}\] addition to alkenes in radical and electrophilic additions reflects a difference in the first species that reacts with the alkene in these addition reactions. In electrophilic addition, \[\ce{H-Br}\] protonates \[\ce{C=C}\] on its least substituted \[\ce{C}\] to give the most highly substituted \[\ce{C+}\]. That carbocation then reacts in a second step with \[\ce{Br+}\] it to end up on the most highly substituted \[\ce{C}\].
Electrophilic addition: \[\ce{R2C=CH2 ->[H-Br] R2C-CH3 ->[Br-] R2CBr-CH3}\]
Radical addition: \[\ce{R2C=CH2 ->[Br^{.}] R2C^{.}-CH2Br ->[H-Br] R2C-CH2Br + Br^{.}}\]
In radical addition, the first species also adds to \[\ce{C=C}\] at the least substituted \[\ce{C}\] to give the most highly substituted \[\ce{C^{.}}\], but that first species to add is \[\ce{Br^{.}}\]. As a result, after the intermediate \[\ce{C^{.}}\] radical abstracts an \[\ce{H}\] from \[\ce{H-Br}\], the \[\ce{Br}\] is on the least substituted \[\ce{C}\].