alkane
Alkane
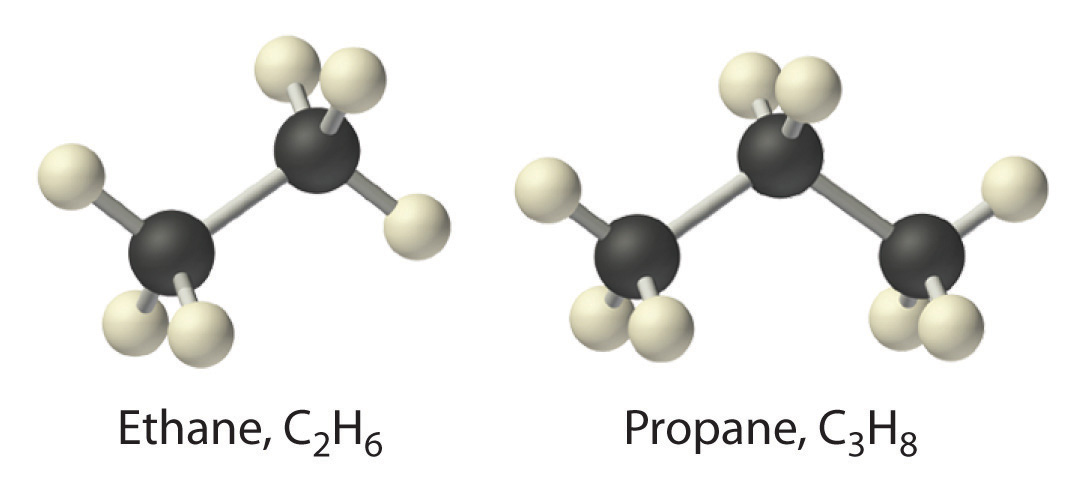
An alkane is an acyclic (linear structure rather than a cyclic structure like benzene) saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which all the carbon-carbon bonds are single. In terms of IUPAC definitions, alkane is defined as acyclic branched or unbranched hydrocarbons having the general formula \[\ce{C_{n}H_{2n+2}}\], and therefore consisting entirely of hydrogen atoms and saturated carbon atoms. In an alkane, each carbon atom is sp3-hybridised with 4 sigma bonds (either \[\ce{C-C}\] or \[\ce{C-H}\]), and each hydrogen atom is joined to one of the carbon atoms (in a \[\ce{C-H}\] bond).
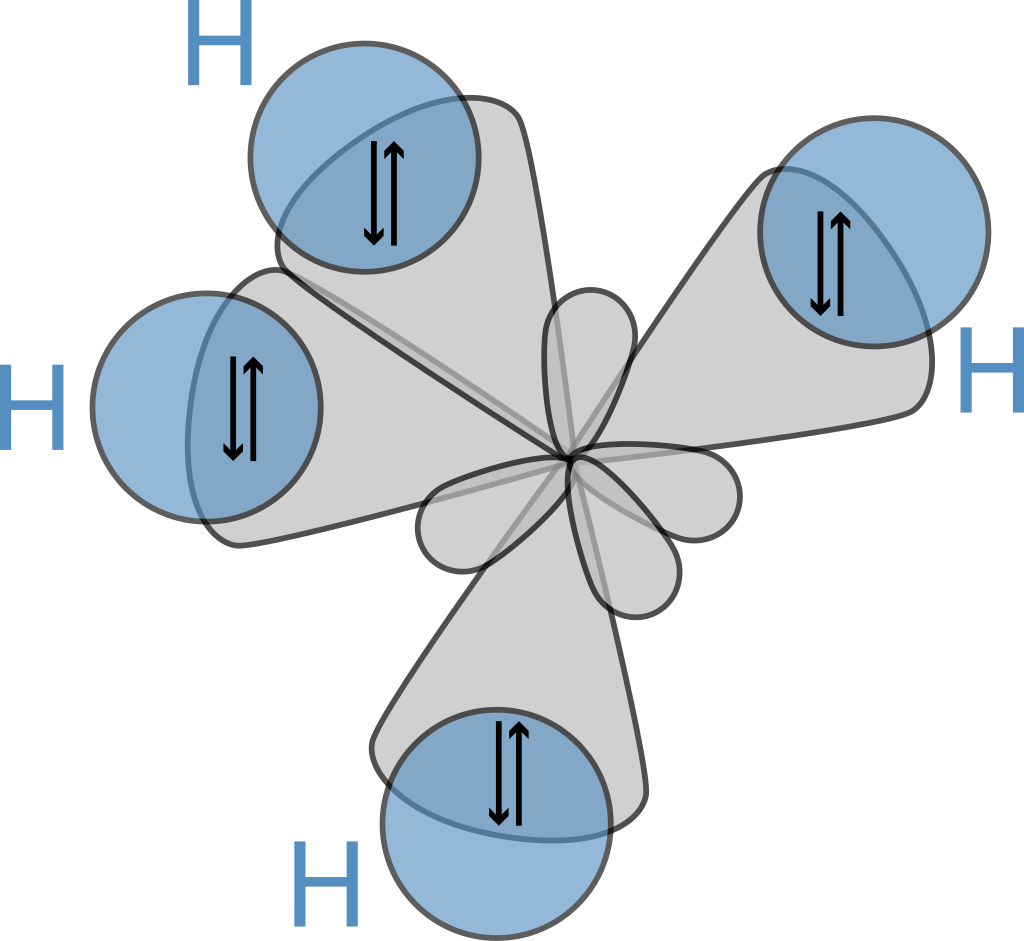
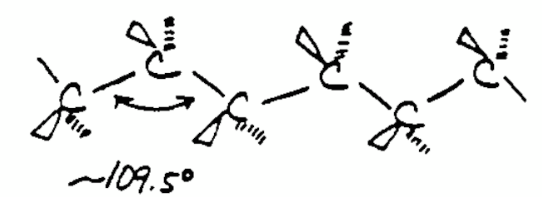
An alkane has only \[\ce{C-H}\] and \[\ce{C-C}\] single bonds. The former result from the overlap of an sp3 orbital of carbon with the 1s orbital of a hydrogen; the latter by the overlap of two sp3 orbitals on adjacent carbon atoms. The bond lengths amount to \[1.09 \times 10^{-10}\text{m}\] for a \[\ce{C-H}\] bond and \[1.54 \times 10^{-10}\text{m}\] for a \[\ce{C-C}\] bond. The spatial arrangement of the bonds is similar to that of the four sp3 orbitals, they are tetrahedrally arranged, with an angle of \[109.47^{\circ}\] between them.
Cycloalkane
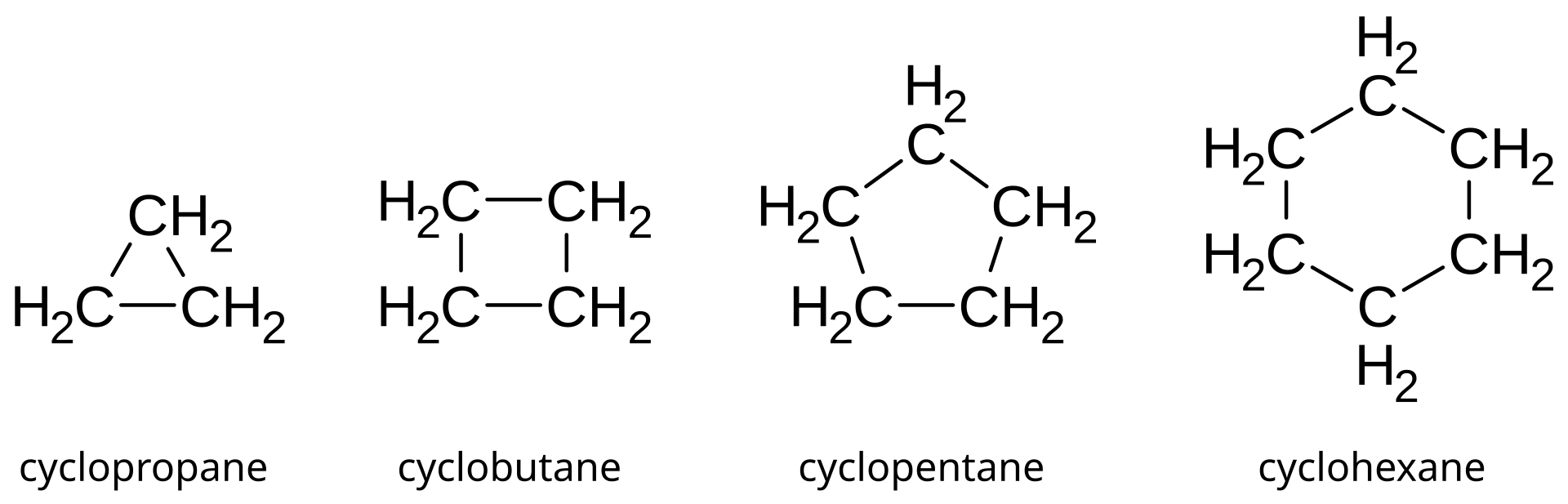
Cycloalkanes are the monocyclic saturated hydrocarbons. In other words, a cycloalkane consists only of hydrogen and carbon atoms arranged in a structure containing a single ring, and all of the carbon-carbon bonds are single. All cycloalkanes are isomers of alkenes. Their boiling points that are also approximately 20 Kelvin higher than the corresponding straight chain alkane.
Example: 2-bromo-1-chloro-3-methylcyclopentane
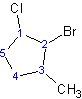
Isomerism
.png) \[\ce{C_{4}}\] alkanes (top) and cycloalkanes (bottom):
\[\ce{C_{4}}\] alkanes (top) and cycloalkanes (bottom):
Alkanes with more than three carbon atoms can be arranged in various ways, forming structural isomers. The simplest isomer of an alkane is the one in which the carbon atoms are arranged in a single chain with no branches. This isomer is sometimes called the n-isomer.
For isomers, the more branched the chain, the lower the boiling point tends to be. Van der Waals dispersion forces are smaller for shorter molecules and only operate over very short distances between one molecule and its neighbors. It is more difficult for short, fat molecules (with lots of branching) to lie as close together as long, thin molecules.
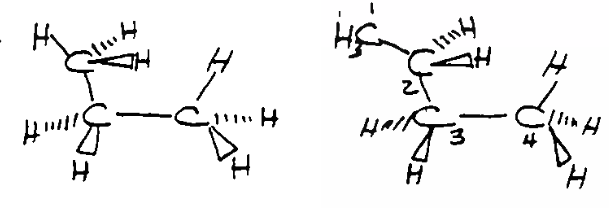 Eclipsed conformation of propene and \[\ce{C3-C4}\] of butene
Eclipsed conformation of propene and \[\ce{C3-C4}\] of butene
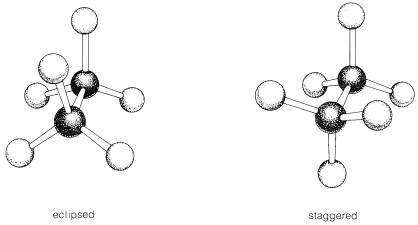 Eclipsed and staggered conformations.
Eclipsed and staggered conformations.
The spatial arrangement of the C-C and C-H bonds are described by the torsion angles of the molecule is known as its conformation. In ethane, the simplest case for studying the conformation of alkanes, there is nearly free rotation about a \[\ce{C-C}\] single bond. Two limiting conformations are important, eclipsed and staggered conformation. The staggered conformation is lower in energy (more stable) than the eclipsed conformation. In highly branched alkanes, the bond angle may differ from the optimal value to accommodate bulky groups. Such distortions introduce a tension in the molecule, known as strain which substantially increases reactivity.
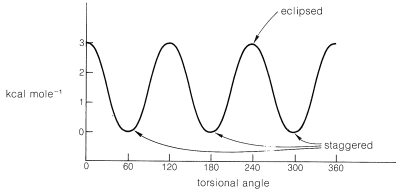 Potential-energy curve for rotation about the \[\ce{C-C}\] bond in ethane
Potential-energy curve for rotation about the \[\ce{C-C}\] bond in ethane
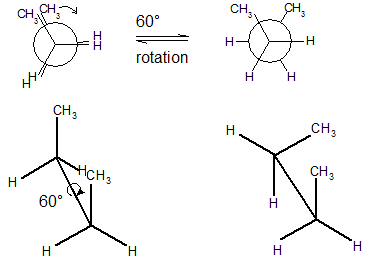 Rotation of the \[\ce{C2-C3}\] single bond in butane
Rotation of the \[\ce{C2-C3}\] single bond in butane
Physical properties of alkane
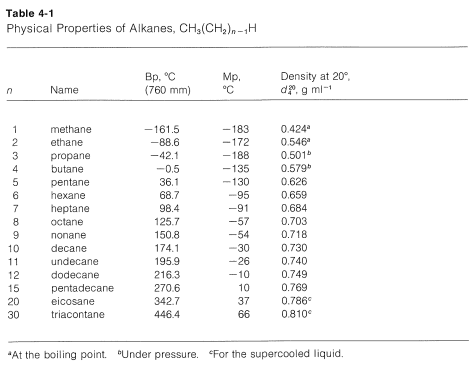
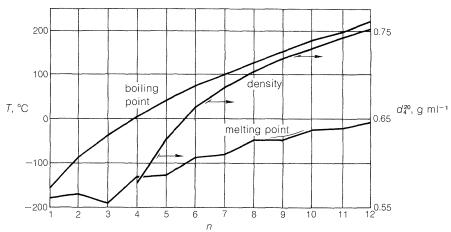
There is not a significant electronegativity difference between carbon and hydrogen, thus, there is not any significant bond polarity. The molecules themselves also have very little polarity. A totally symmetrical molecule like methane is completely non-polar, meaning that the only attractions between one molecule and its neighbors will be van der Waals dispersion forces. These forces will be very small for a molecule like methane but will increase as the molecules get bigger. Therefore, the boiling points of the alkanes increase with molecular size.
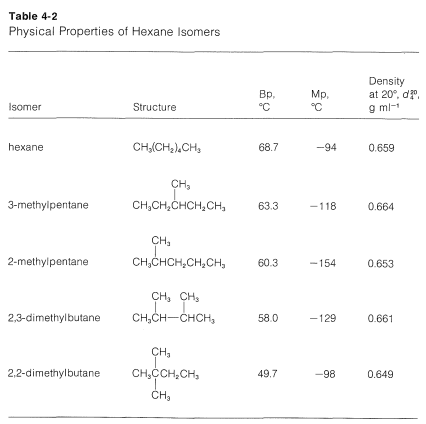
This trend generally will be true, except when the number of carbons is small and when the hydrocarbon chain has polar substituents. Consider compounds such as alcohols, \[\ce{ROH}\], which have polar \[\ce{O-H}\] groups. Polarity causes molecules to associate with one another, which decreases their volatility, raises melting points, increases solubility in polar liquids, and decreases solubility in nonpolar liquids. Which explains why methanol, \[\ce{CH3OH}\], is much less volatile and much more water-soluble than methane, \[\ce{CH4}\]. But we find that the water-solubility of alcohols falls off rapidly with the length of the carbon chain, certainly faster than expected for a simple homologous series effect. Whereas methanol, \[\ce{CH3OH}\], and ethanol, \[\ce{C2H5OH}\], are completely soluble in water, butanol, \[\ce{C4H9OH}\], is only slightly soluble.
Chemical properties of alkane
Acid-base behaviour
The acid dissociation constant (\[\text{p}K_{a}\]) values of all alkanes are estimated to range from 50 to 70, depending on the extrapolation method, hence they are extremely weak acids that are practically inert to bases. They are also extremely weak bases, undergoing no observable protonation in pure sulfuric acid.
Combustion reaction
All alkanes react with oxygen in a combustion reaction, although they become increasingly difficult to ignite as the number of carbon atoms increases. The general equation for complete combustion is:
\[\ce{C_{$n$}H_{$2n+2$} + $\left(\frac{3}{2}n+\frac{1}{2}\right)$ O2 -> $n$ CO2 + $(n+1)$H2O}\]
This reaction requires an initial energy input to overcome the activation energy barrier, allowing the formation of free radicals that propagate the combustion process.
In the absence of sufficient oxygen, carbon monoxide or even soot can be formed, as shown below:
\[\ce{C_{$n$}H_{$2n+2$} + $\left(n+\frac{1}{2}\right)$O2 -> $(n+1)$H2O + $n$CO}\] and \[\ce{C_{$n$}H_{$2n+2$} + $\left(\frac{1}{2}n+\frac{1}{2}\right)$O2 -> $(n+1)$H2O + $n$C}\]
To simplify this, an example would be the combustion of butane, \[\ce{C4H10}\]
- \[\ce{2C4H10 + 13O2 -> 8CO2 + 10H2O}\] (complete combustion)
- \[\ce{2C4H10 + 9O2 -> 8CO + 10H2O}\] (incomplete combustion producing carbon monoxide)
- \[\ce{2C4H10 + 5O2 -> 8C + 10H2O}\] (incomplete combustion producing soot)
Reactions
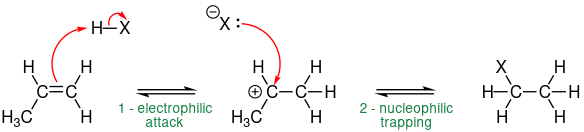
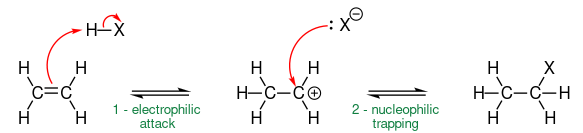
Hydrogenation reaction by electrophilic addition
Alkene reacts with hydrogen in the presence of Pt/Ni catalyst of temperature about 150 Celsius, i.e. \[\ce{CH2=CH2 + H2 ->[\text{Pt/Ni}][\Delta] CH3-CH3}\].
Cracking longer chain alkanes
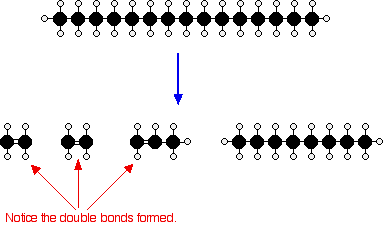
\[\ce{C15H32 ->[\ce{Al2O3}][\Delta 500^{\circ}\,\text{C}] 2C2H4 + C3H6 + C8H18}\]. Cracking longer chain alkanes requires a catalyst, usually aluminium oxide (zeolites), \[\ce{Al2O3}\] and a temperature of about 500 Celsius.
Free-radical substitution of alkanes
Alkanes undergo free-radical substitution in the presence of \[\ce{Cl2}\] and \[\ce{Br2}\] alongside UV light.