redox reactions
Redox reactions
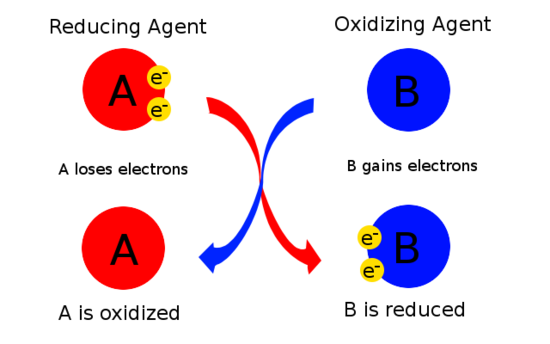
Oxidation
There are multiple definitions to oxidation:
- Gain of oxygen, e.g. \[\ce{CO}\] in \[\ce{Fe2O3 + 3CO -> 2Fe + 3CO2}\]
- Loss of hydrogen, e.g. \[\ce{CH3CH2OH + [O] -> CH3CHO + H2O}\], where \[\ce{[O]}\] refers to the oxygen provided by an oxidising agent such as \[\ce{Cr2O7^{2-}}\]
- Loss of electrons, e.g. \[\ce{Mg}\] in \[\ce{CuO + Mg -> Cu + MgO}\], which can also be written as \[\ce{Cu^{2+} + Mg -> Cu + Mg^{2+}}\]
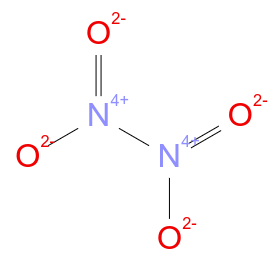
If a substance is an oxidising agent, then its role in a chemical reaction is to oxidise another substance, meaning it will gain electrons while the other will lose electrons. Oxidants are usually chemical substances with elements in high oxidation states (large positive oxidation numbers) such as \[\ce{N2O4}\]:
or else highly electronegative elements, such as \[\ce{F2}\] and \[\ce{I2}\].
The reason oxidation having two meanings is that the term oxidation orginally implied a reaction with oxygen to form an oxide. Ultimately, the meaning was generalized to include all processes involving the loss of electrons or the increase in the oxidation state of a chemical species.
Reduction
The defitions to reduction is as follows
- Loss of oxygen, e.g. \[\ce{Fe}\] is \[\ce{Fe2O3 + 2Al -> Al2O3 + 2Fe}\]
- Gain of hydrogen
- Gain of electrons
If a substance is a reducing agent, it's role in a chemical reaction is to reduce another substance while it itself will be oxidised, e.g. a reducing agent will donate electrons to another substance, reducing it. Reductants are very diverse, but usual reductants are electropositive element metals such as \[\ce{Li}\], \[\ce{Na}\], \[\ce{Mg}\], \[\ce{Fe}\], \[\ce{Zn}\] and \[\ce{Al}\].
Disproportionation
Disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation state.
An example reaction would be phosphorous acid heated to 200°C to give phosphoric acid and phosphine: \[\ce{4H3PO3 -> 3H3PO4 + PH3}\].
Here, \[\ce{P}\]'s original oxidation is +4 (see here), which then gets oxidised to both +5 in \[\ce{H3PO4}\] and 0 in \[\ce{PH3}\].
Balancing redox reactions
Let's take \[\ce{MnO4- + I- -> I2 + Mn^{2+}}\] as an example.
Seperate the half-reactions
The equation above can be separated into \[\ce{MnO4- -> Mn^{2+}}\] (reduced from +7 to +2) and \[\ce{I- -> I2}\] (oxidised from -1 to 0).
Balancing the all atoms other than hydrogen and oxygen
\[\ce{I- -> I2}\] should be balanced to become \[\ce{2 I- -> I2}\], meanwhile the manganese half equation is already balanced.
Balancing the oxygen and hydrogen atoms
\[\ce{MnO4- -> Mn^{2+} + 4 H2O}\], balancing the four oxygen atoms on the left-hand side of the equation. Now we have an extra 8 hydrogen atoms on the right-hand side of the equation to balance the hydrogen atoms out.
Therefore, \[\ce{MnO4- + 8 H+ -> Mn^{2+} + 4 H2O}\].
Balancing the electrons
\[\ce{2 I- -> I2}\] has an overall charge of -2 on the left side, therefore, \[\ce{2 I- -> I2 + 2 e-}\].
\[\ce{MnO4- + 8 H+ -> Mn^{2+} + 4 H2O}\], here, the overall charge of the left-hand side of the equation is ((-1) + (+8)), due to \[\ce{MnO4-}\] being -1 and 8 hydrogen ions, which gives us an overall charge of +7. On the right-hand side of of the equation, the overall charge is +2, as water molecules have zero overall charge. Therefore,
Multiply both equations by a common multiple to allow both of their \[\ce{e-}\] to cancel out
\[\ce{2 I- -> I2 + 2 e-}\] becomes \[\ce{10 I- -> 5 I2 + 10 e-}\], while \[\ce{MnO4- + 8 H+ + 5 e- -> Mn^{2+} + 4 H2O}\] becomes \[\ce{2 MnO4- + 16 H+ + 10 e- -> 2 Mn^{2+} + 8 H2O}\].
Combining both equations and cancelling out the \[\ce{e-}\] we get, \[\ce{10 I- + 2 MnO4- + 16 H+ -> 5 I2 + 2 Mn^{2+} + 8 H2O}\].