free radical
Free radical
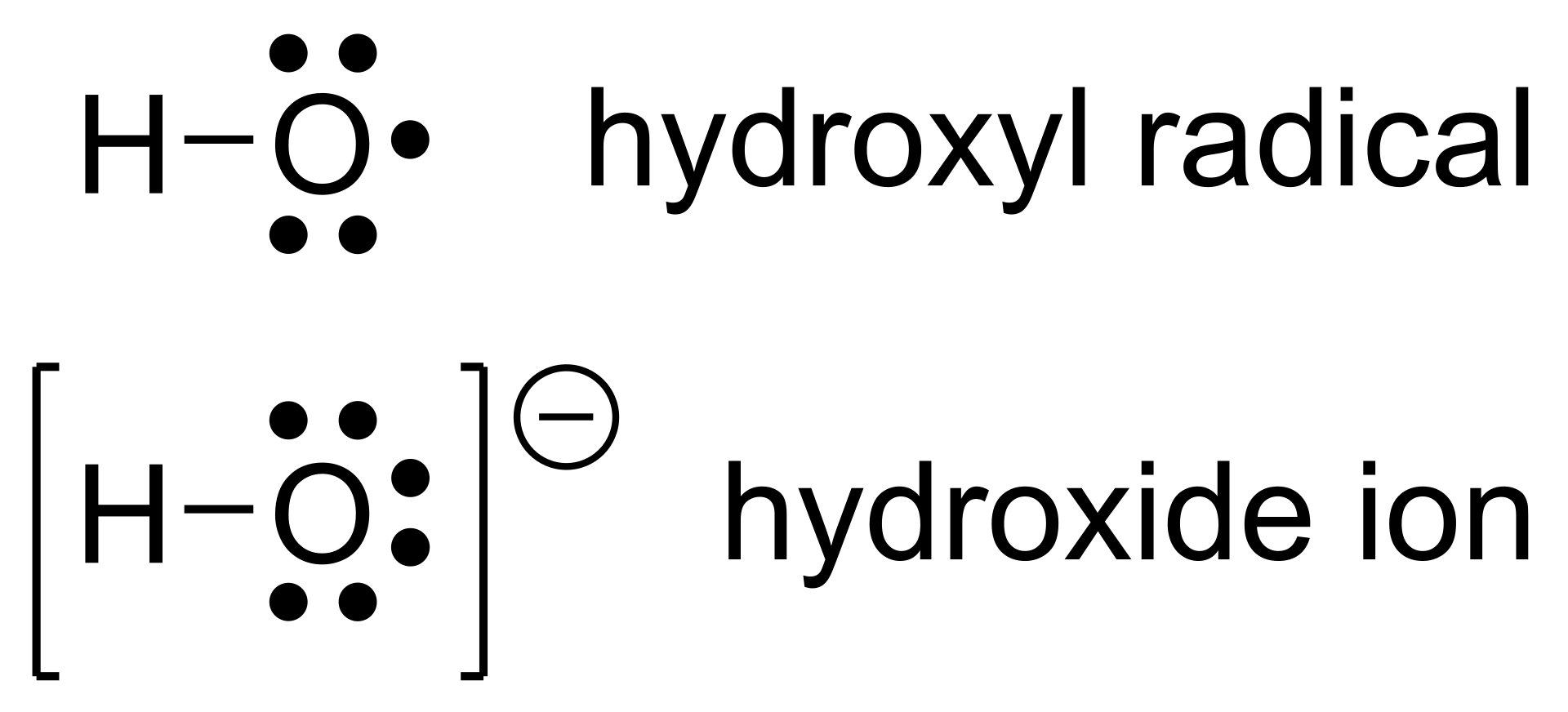
A free radical, also known as a radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize (join with another free radical to form one bigger and more stable molecule).
In chemical equations, free radicals are frequently denoted by a dot placed immediately to the right of the atomic symbol or molecular formula as follows: \[\ce{Cl2 ->[UV] Cl^{.} + Cl^{.}}\]
Stability
Organic radicals
Radicals are molecules that have an unpaired electron, making them highly reactive. Their stability depends on two main aspects: thermodynamic stability (how low in energy the radical is) and kinetic stability or persistency (how long the radical exists before reacting further).
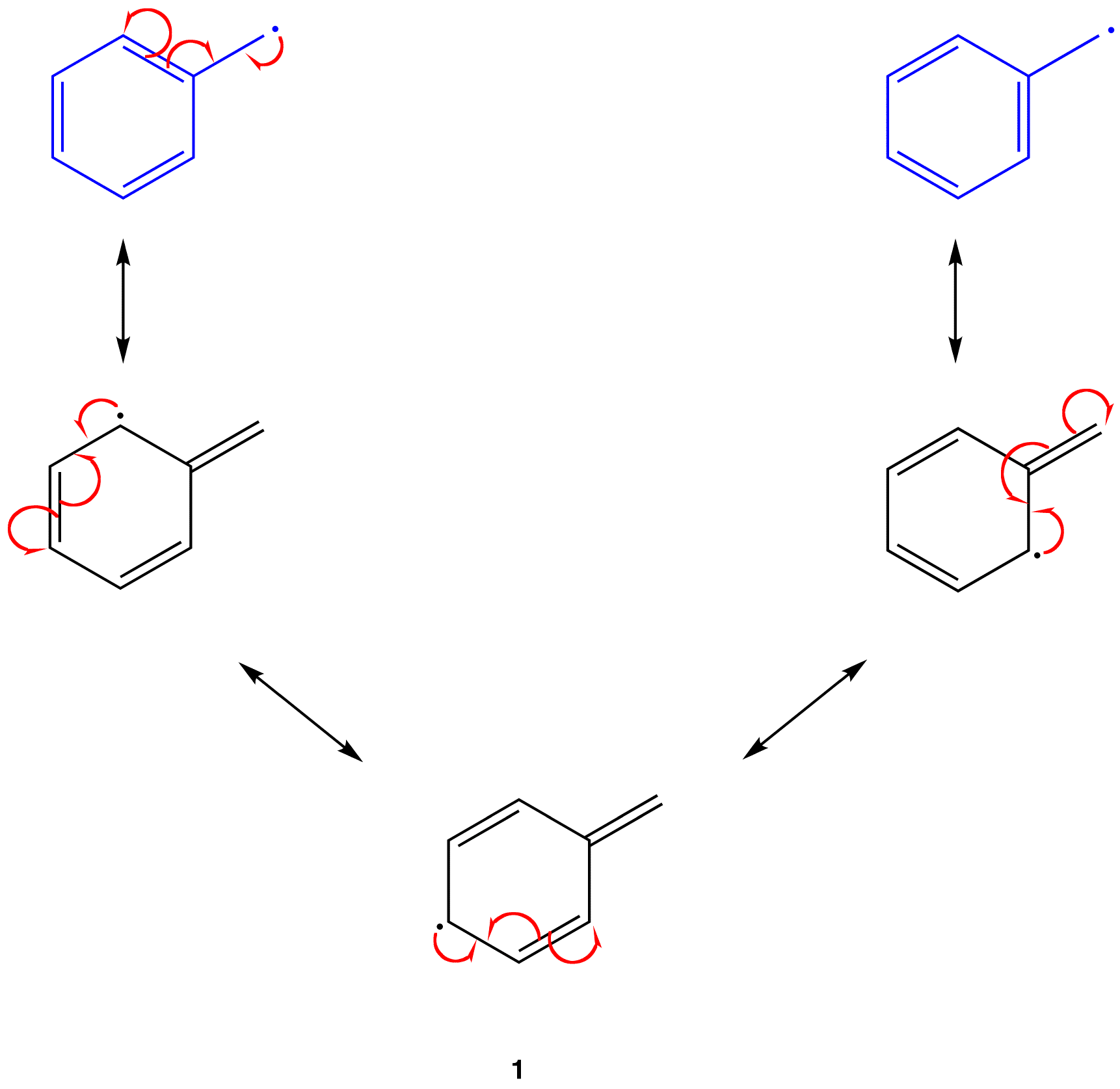
These two types of stability don't always align. For example, benzylic radicals (radicals located next to a benzene ring) are thermodynamically stable because their unpaired electron can spread out through the ring (resonance). However, they don't last long because they quickly pair up with another radical to form a stable dimer. The term "stabilized" are for radicals with weaker bonds and "persistent" for those that live longer.
Electronegativity
In radicals, the more electronegative the atom holding the unpaired electron, the less stable the radical becomes. This is because highly electronegative atoms hold onto their electrons tightly, making it harder for the radical to stabilize. For instance, a radical on an oxygen atom, \[\ce{O^{.}}\], is less stable than one on a carbon atom, \[\ce{C^{.}}\].
Additionally, the hybridisation of the carbon affects stability: radicals on sp3-hybridized carbons (25% s-character, which is the percentage contribution of the s orbital in the hybridization process) are more stable than those on sp-hybridized carbons (50% s-character) because the electrons in sp-hybridized orbitals are held closer to the nucleus, making the radical less stable.
Delocalization and resonance
Delocalization refers to the spreading out of an unpaired electron over multiple atoms within a molecule. This can happen through resonance, where the radical can be represented by multiple structures with the unpaired electron in different positions.
Delocalization makes radicals more stable because the electron density is not concentrated on a single atom. For example, the radical formed from benzyl chloride, \[\ce{C6H5CH2Cl}\] can spread its unpaired electron over the aromatic ring, making it more stable than a radical without such delocalization.
Reactions involving free radicals

Chain reactions involving free radicals can usually be divided into three distinct processes. These are initiation, propagation, and termination. The initiation phase describes the step that initially creates a radical species. In most cases, this is a homolytic cleavage event, and takes place very rarely due to the high energy barriers involved. Often the influence of heat, UV radiation, or a metal-containing catalyst is necessary to overcome the energy barrier.
The propagation phase describes the chain part of chain reactions. Once a reactive free radical is generated, it can react with stable molecules to form new free radicals. These new free radicals go on to generate yet more free radicals, and so on. Propagation steps often involve hydrogen abstraction or addition of the radical to double bonds.
Chain termination occurs when two free radical species react with each other to form a stable, non-radical species. Although this is a very thermodynamically downhill (exothermic) event, it is also very rare due to the low concentration of radical species and the small likelihood of two radicals colliding with one another.