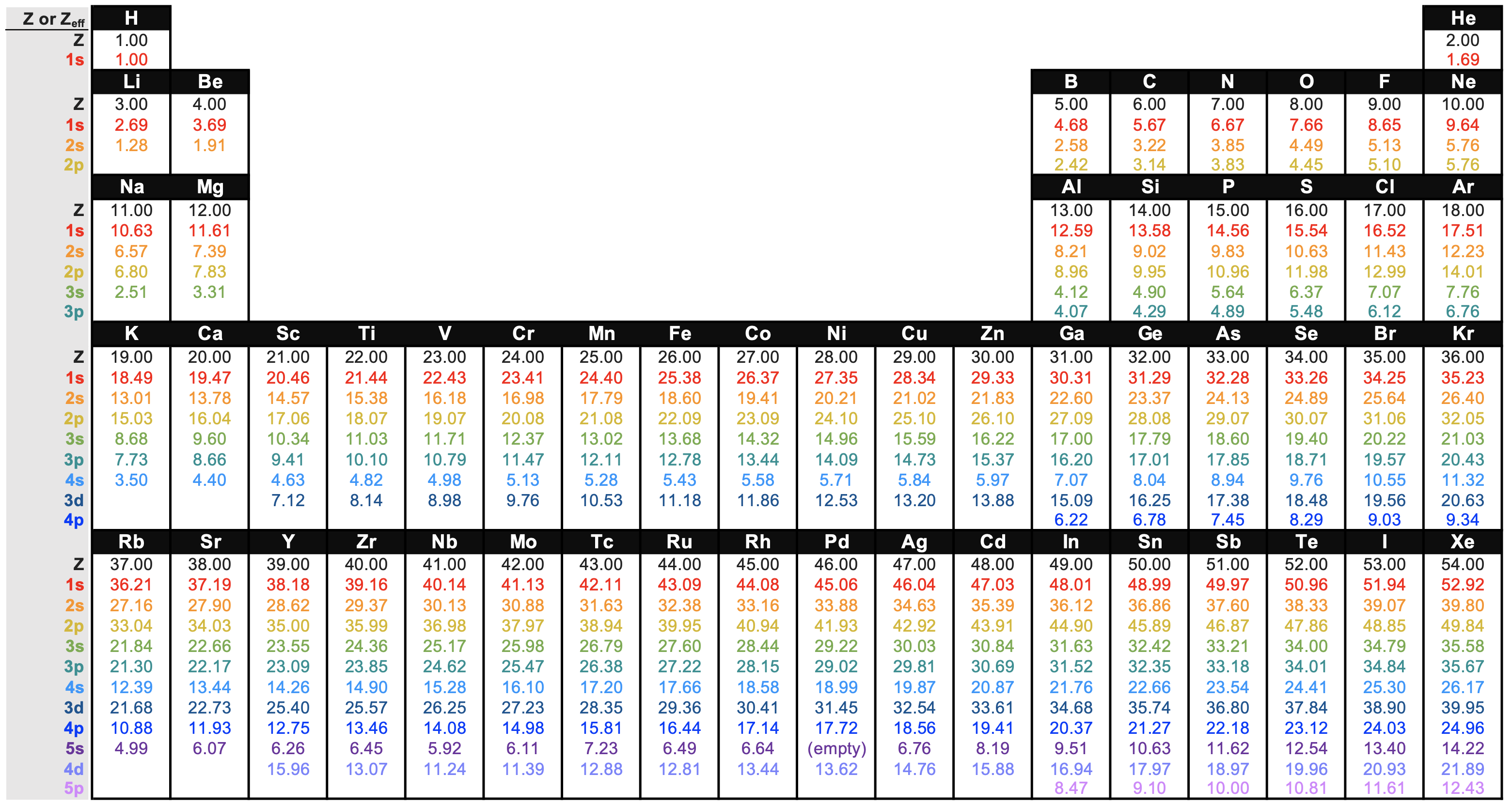
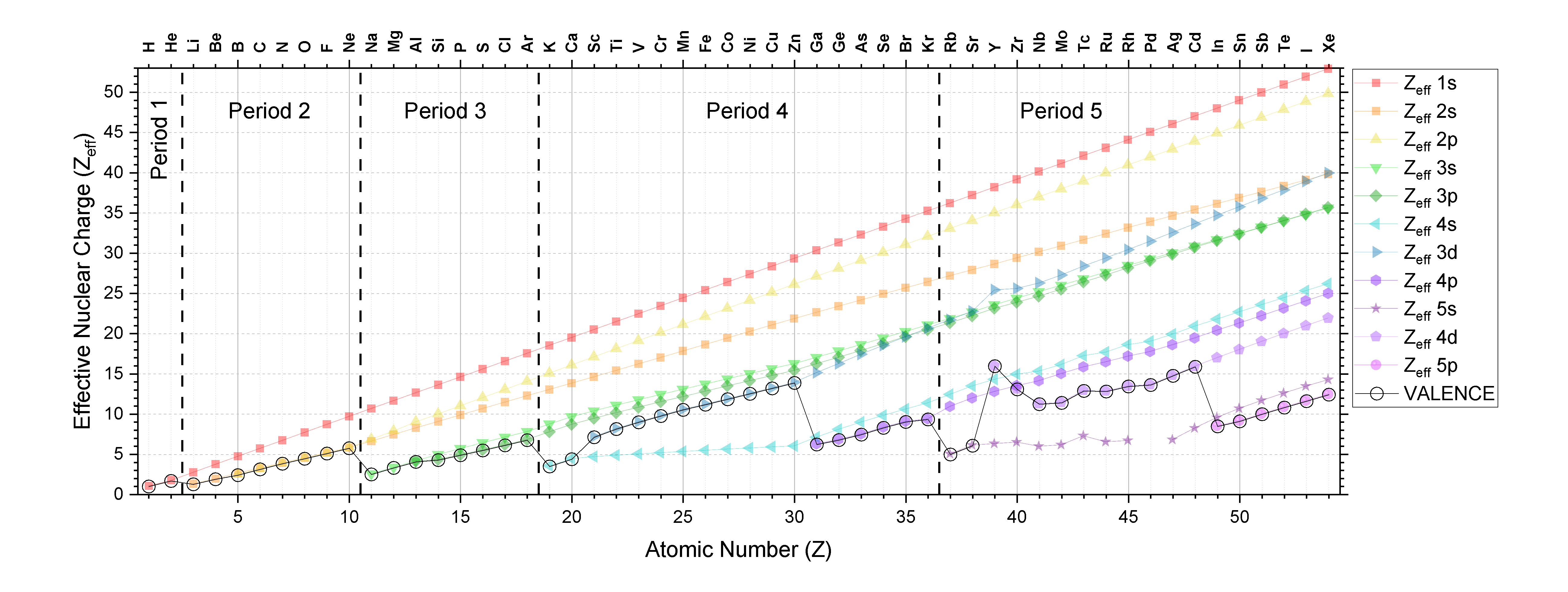
effective nuclear charge
Effective nuclear charge
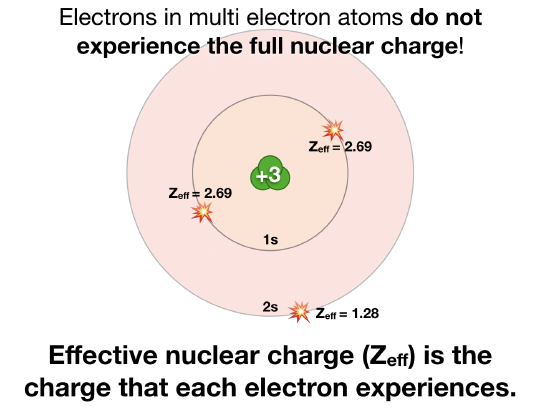
Effective nuclear charge, commonly denoted as \[Z_{\textit{eff}}\] is the actual amount of positive charge experienced by an electron in a multi-electron atom. Electrons don't experience the full charge exerted by the nucleus due to:
- Size of atom
-
Electron shielding
-
Orbital penetration
A higher \[Z_{\textit{eff}}\] corresponds to a lower potential energy as \[\text{Potential energy}=-\frac{kZe^{2}}{r}\], where \[k\] is Coulomb's constant, \[Z\] is the number of protons in the atom, \[r\] is the radius of separation (distance between electron and nucleus) while \[e\] is one unit of charge. From the formula we can see that the smaller \[r\] is the more negative the potential energy is. In layman terms, a more negative potential energy = lower potential energy and more tightly bound to the nucleus as you would need the same amount of energy to remove it from the nucleus.
The reason why potential energy is negative is because of this:
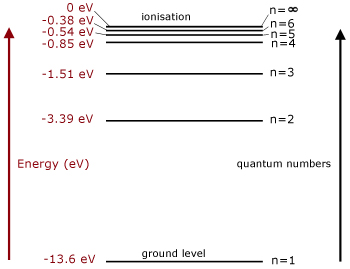
Orbital energies are measured relative to an electron at infinity (i.e. one which has been ionized), which corresponds to an energy of 0 eV. An electron which is lower in energy than this (i.e. bound to an atom) has a negative energy. The first six energy levels for hydrogen are shown above. As you can see, the energies converge to 0 as n, the principal quantum number, goes to infinity. In order to ionize an electron it must be given enough energy to promote it to the \[n=\infty\] energy level. Therefore the higher the energy of the electron, the less energy is required to ionize it.
Quote: A negative potential energy means that work must be done against the electric field in moving the charges apart!
For a many electron atom, \[Z_{\textit{eff}}\] for each orbital can be roughly calculated with Slater's rules.
The approximation for \[Z_{\textit{eff}}\] is given as the below: