SN2 reaction
SN2 reaction
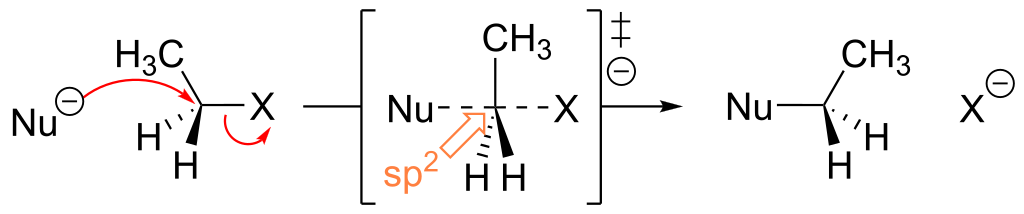

The bimolecular nucleophilic substitution (SN2) is a type of reaction mechanism that is common in organic chemistry. In the SN2 reaction, a strong nucleophile forms a new bond to an sp3-hybridised carbon atom via a backside attack, all while the leaving group detaches from the reaction centre in a concerted (i.e. simultaneous) fashion.
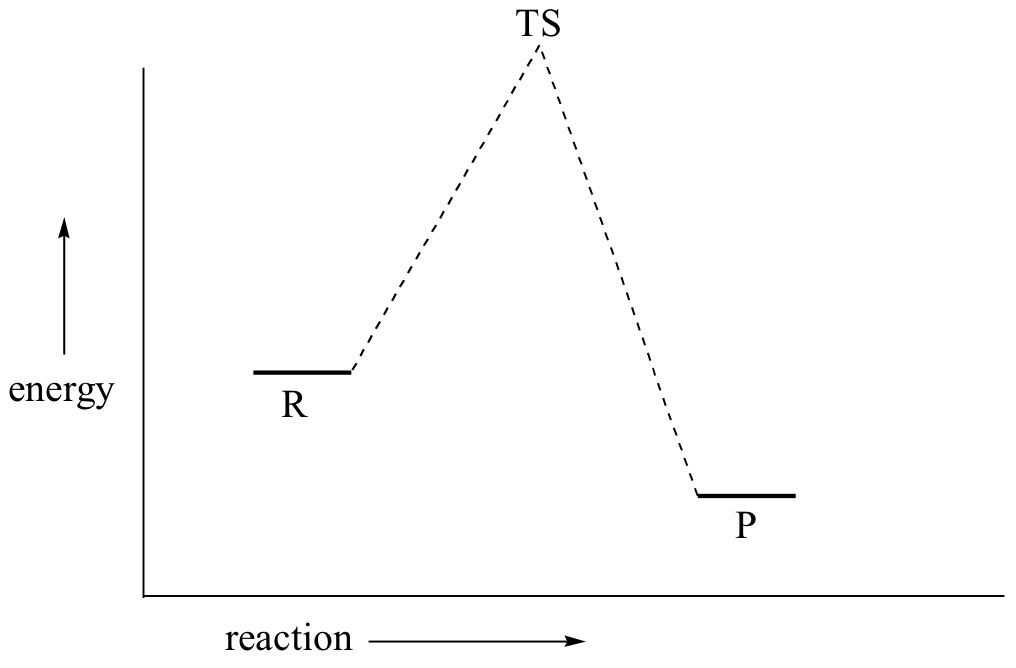
The reason why it's called an SN2 mechanism is that S stands for substitution, N stands for nucleophilic and 2 refers to the fact that this is a bimolecular reaction, i.e. the overall rate depends on a step in which two separate molecules collide. A potential energy diagram for this reaction shows the transition state (TS) as the highest point on the pathway from reactants to products.
Leaving group
In order to understand the nature of the leaving group, it is important to first discuss factors that help determine whether a species will be a strong base or weak base. A Lewis base is defined as a species that donates a pair of electrons to form a covalent bond.
Generally, if we move from the left to right of the periodic table, electronegativity increases. As it increases, basicity will decrease, meaning a species will be less likely to act as base, i.e., the species will be less likely to share its electrons. Similarly, as we move from top to bottom of the periodic table, the size of the atom increases. With an increased size, basicity decreases. Additionally, resonance stabilises a structure, thus making the species less willing to share its electrons. Therefore, resonance stabilised structures are weak bases.
Now that we've established that, it makes sense that weak bases make the best leaving groups, as for it to leave, it must be able to accept electrons.
Mechanism
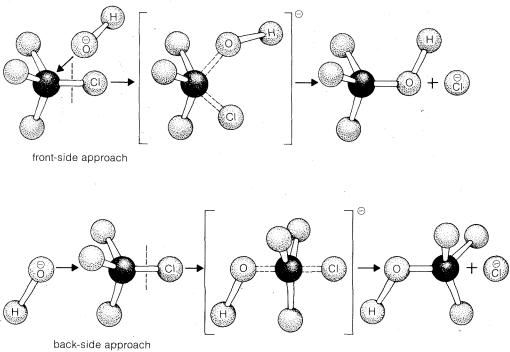
If you look carefully at the progress of the SN2 reaction, the nucleophile, being an electron-rich species, must attack the electrophilic carbon from the back side relative to the location of the leaving group. Approach from the front side simply doesn't work: the leaving group, which is also an electron-rich group blocks the way. The result of this backside attack is that the stereochemical configuration at the central carbon inverts as the reaction proceeds. In a sense, the molecule is turned inside out.

At the transition state, the electrophilic carbon and the three "R" substituents all lie on the same plane.
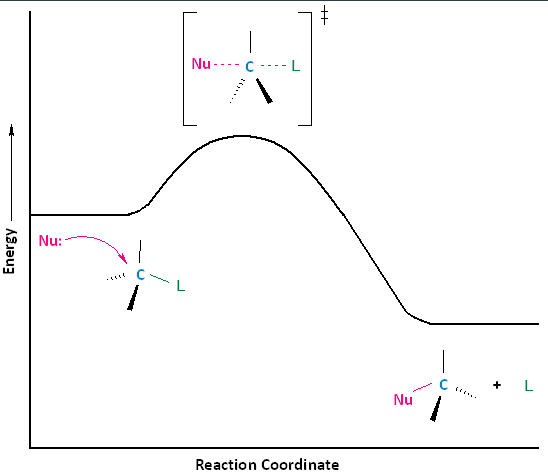
SN2 reactions are also said to be concerted, i.e. they are a one step process. This means that the process whereby the nucleophile attacks and the leaving group leaves is simultaneous. Hence, the bond-making between the nucleophile and the electrophilic carbon occurs at the same time as the bond-breaking between the electophilic carbon and the halogen. Thus, the transition state formed right after a nucleophilic attack is a very short-lived species that cannot be isolated or directly observed.
Steric hindrance
Naturally, as each hydrogen is replaced by an \[\ce{R}\] group, the rate of reaction is significantly diminished. This is because the addition of one or two \[\ce{R}\] groups shields the backside of the electrophilic carbon, impeding nucleophilic attack. Which we can summarise as \[3^{\circ}<2^{\circ}<1^{\circ}\,\text{and methyl}\].
Stereospecific
The SN2 reaction is stereospecific, i.e. different stereoisomers react to give different stereoisomers of the product.