physical properties of group 2 elements
Physical properties of group 2 elements
Atomic radius
The increasing atomic radius can be explained by:
- increasing number of layers of electrons around the nucleus
- decreased pull towards the outer electrons from the nucleus
Ionic radius
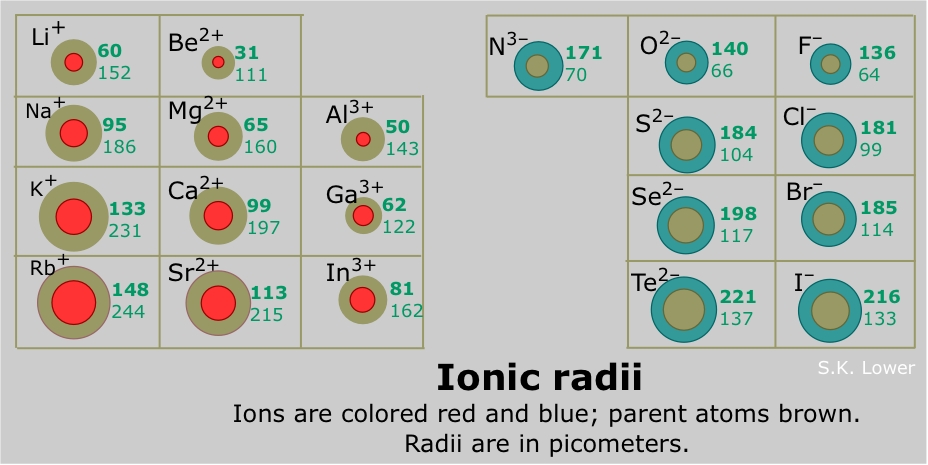
Similar to the trend for increasing atomic radius,
- Each added shell increases the average distance of the outermost electrons from the nucleus, thus increasing the overall size of the ion
- Increase in the number of inner electron shells also increases the shielding effect, decreasing effective nuclear charge allowing the outer electrons to be less tightly held by the nucleus
Ionisation energy
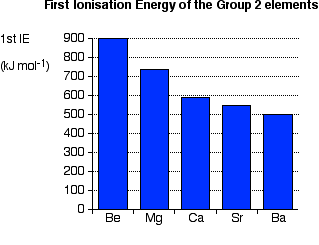
The distance between the nucleus and the outer electrons increases and so they become easier to remove, thus the ionisation energy decreases.
Electronegativity
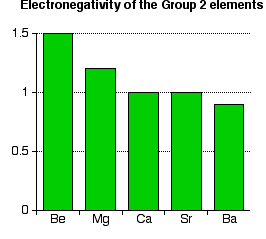
Increase in atomic size and shielding effect weakens the attraction between the nucleus and the valence electrons (decreased effective nuclear charge), making them less effective at attracting electrons from other atoms when forming bonds
Referenced by:
No backlinks found.