nucleophile
Nucleophile
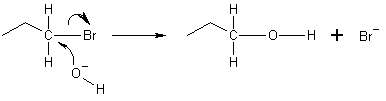
The image above shows a hydroxide ion acting as a nucleophile, converting a haloalkane into an alcohol.
A nucleophile is a chemical species that forms bonds by donating an electron pair. Because nucleophiles donate electrons, they are Lewis bases. The term nucleophilic describes the readiness of a nucleophile to bond with positively charged atomic nuclei.
In both laboratory and biological organic chemistry, the most relevant nucleophilic atoms are oxygen, nitrogen, and sulfur, and the most common nucleophilic functional groups are water, alcohols, phenols, amines, thiols, and occasionally carboxylates. In laboratory reactions, halide and azide anions are also commonly seen acting as nucleophiles. Carbon atoms can also be nucleophiles, with the cyanide ion, \[\ce{(C#N)-}\], being the best example.
Horizontal trends in nucleophilicity
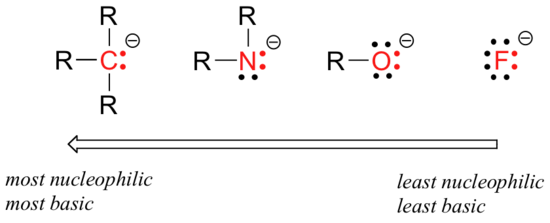
The reasoning behind the horizontal nucleophilicity trend is that more electronegative elements hold their electrons more tightly, and are less able to donate them to form a new bond.
Vertical trends and effects of solvents in nucleophilicity
Let's stake a simple example of the SN2 reaction:
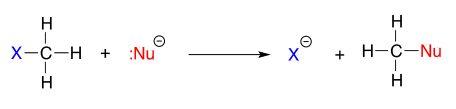
where \[\ce{Nu-}\] is one of the halide ions and \[\ce{X}\] is a common leaving group.
Let's start with the case with protic solvents, i.e. a solvent that has a hydrogen bonded to an oxygen or nitrogen such as water, methanol and ethanol. The reaction will go fastest when iodide is the nucleophile, and slowest when fluoride is the nucleophile, reflecting the relative strength of the nucleophile.
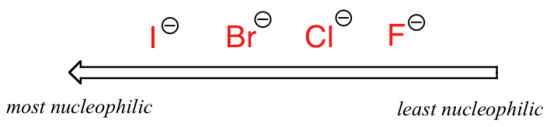
The reason why is that protic solvent molecules form very strong ion-dipole interactions with the negatively charged nucleophile, essentially creating a "solvent cage" around the nucleophile.
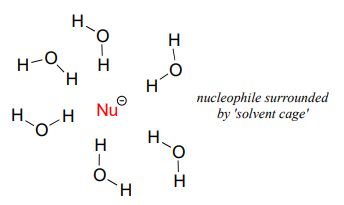
In order for the nucleophile to attack, it must break free, at least in part, from its solvent cage. The lone pair electrons on the larger, less basic iodide ion interact less tightly with the protons on the protic solvent molecules, thus the iodide nucleophile is better able to break free from its solvent cage compared the smaller, more basic fluoride ion, is more highly shielded by stronger interactions with the solvent molecules. The electron cloud of the fluoride ion is smaller and much less polarizable than that of an iodide ion.
Now, this is not the case when we are talking about a polar aprotic solvent such as acetone, in which there is a molecular dipole but no hydrogens bound to oxygen or nitrogen.
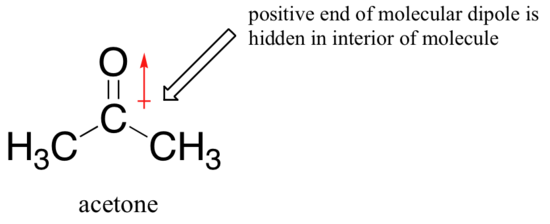
Now, fluoride is the best nucleophile, and iodide the weakest. The reason for the reversal is that, with an aprotic solvent, the ion-dipole interactions between solvent and nucleophile are much weaker. The positive end of the solvent's dipole is hidden in the interior of the molecule, and thus it is shielded from the negative charge of the nucleophile.
A weaker interaction between the solvent and nucleophile means a weaker solvent cage for the nucleophile to break through, so the solvent effect is much less important, and the more basic fluoride ion is also the better nucleophile.
Resonance effects on nucleophilicity

Resonance effects also come into play when comparing the nucleophilicity of different molecules. If the electron lone pair on a heteroatom is delocalized by resonance, it is inherently less reactive, thus less nucleophilic. An alkoxide ion, for example, is more nucleophilic than a carboxylate group, even though in both cases the nucleophilic atom is a negatively charged oxygen. In the alkoxide, the negative charge is localized on a single oxygen, while in the carboxylate the charge is delocalized over two oxygen atoms by resonance.

Another instance of such effects is found in the nitrogen atom on an amide, which is less nucleophilic than the nitrogen of an amine, due to the resonance stabilization of the nitrogen lone pair provided by the amide carbonyl group.
Steric hindrance and nucleophilicity
Take the example of 2-methylpropan-2-ol and methanol.
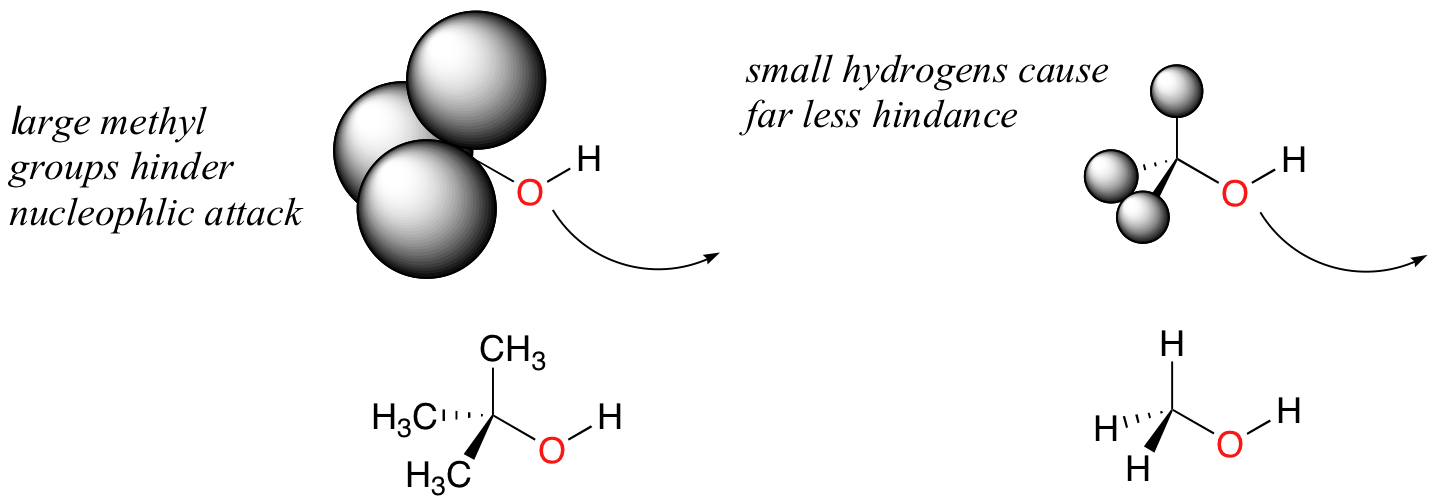
The comparatively bulky methyl groups on the tertiary alcohol effectively block the route of attack by the nucleophilic oxygen, slowing the reaction down considerably.