chemical properties of period 3 elements
Chemical properties of period 3 elements
Reactions with oxygen
Sodium: Forms \[\ce{Na2O}\], \[\ce{4 Na + O2 -> 2 Na2O}\]
Magnesium: Forms \[\ce{MgO}\], \[\ce{2 Mg + O2 -> 2 MgO}\]
Aluminium: Forms \[\ce{Al2O3}\], \[\ce{4 Al + 3 O2 -> 2 Al2O3}\]
Silicon: Forms \[\ce{SiO2}\], \[\ce{Si + O2 -> SiO2}\]
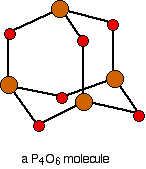
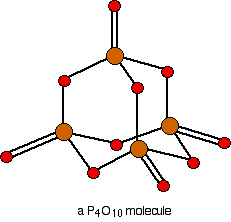
Phosphorus: Forms both Phosphorus(III) oxide, \[\ce{P4O6}\], \[\ce{P4 + 3 O2 -> P4O6}\] and Phosphorus(V) oxide, \[\ce{P4O10}\], \[\ce{P4 + 5 O2 -> P4O10}\]
In case of \[\ce{P4O6}\] only the electrons in the 3p orbital take part. Resulting in formation of 3 bonds with \[\ce{O}\]. But in case of \[\ce{P4O10}\] one of the electrons in the 3s orbital is excited to the vacant 3d orbital. Now it has 5 unpaired electrons and can form 5 bonds therefore forming \[\ce{P4O10}\]. \[\ce{P4}\] is used here as the most reactive form of molecular phosphorous, and is the only molecular phosphorous at standard temperature and pressure.

Sulfur: Forms both Sulfur dioxide, \[\ce{SO2}\], \[\ce{S + O2 -> SO2}\] and Sulfur trioxide, \[\ce{SO3}\], \[\ce{2 SO2 + O2 -> SO3}\]
Reactions with chlorine
Sodium: Forms \[\ce{NaCl}\], \[\ce{2 Na + Cl2 -> 2 NaCl}\]
Magnesium: Forms \[\ce{MgCl2}\], \[\ce{Mg + Cl2 -> MgCl2}\]
Aluminium: Forms \[\ce{AlCl3}\], \[\ce{2 Al + 3 Cl2 -> 2 AlCl3}\], aluminium chloride
Silicon: Forms \[\ce{SiCl4}\], \[\ce{Si + 2 Cl2 -> SiCl4}\], silicon(IV) chloride
Phosphorus: Forms both phosphorus(III) chloride, \[\ce{PCl3}\], \[\ce{P4 + 6 Cl2 -> 4 PCl3}\] and phosphorus(V) chloride, \[\ce{PCl5}\], \[\ce{P4 + 10 Cl2 -> 4 PCl5}\]
Sulfur: Forms disulfur dichloride, \[\ce{S2Cl2}\], \[\ce{2 S + Cl2 -> S2Cl2}\]
Reactions with water
Sodium: Reacts vigorously as water as it loses it's valence electron easily as it is very energetically favourable for the sodium atom. \[\ce{2 Na(s) + 2 H2O(l) -> 2 NaOH(aq) + H2(g)}\].
Magnesium: Reacts less vigorously as the attraction between \[\ce{Mg^{2+}}\] and it's delocalised electrons are much stronger than sodium atoms. However, it still reacts strongly with water when heated strongly in steam: \[\ce{Mg(s) + H2O(g) -> MgO(s) + H2(g)}\].
The trend of decreasing strength in reactions continue down the period:
Aluminium: Aluminium powder heated in steam produces hydrogen and aluminium oxide. The reaction is relatively slow because of the existing strong aluminium oxide layer on the metal, and the build-up of even more oxide during the reaction. \[\ce{2 Al + 3 H2O -> Al2O3 + 3 H2}\].
Silicon: The common shiny grey lumps of silicon with a rather metal-like appearance are fairly unreactive and will only react with steam at red heat to produce silicon dioxide and hydrogen. \[\ce{Si + 2 H2O -> SiO2 + 2 H2}\].
Reactions of oxides with water
Giant ionic structures:
Sodium oxide (very soluble, as the attraction between sodium ion and oxide ions within the crystal lattice is weaker than the attraction between polar water molecules) \[\ce{Na2O + H2O -> 2 NaOH}\];
magnesium oxide (slightly soluble, as the attraction between magnesium ion and oxide ions is stronger), \[\ce{MgO + H2O -> Mg(OH)2}\];
aluminium oxide is insoluble in water as the strength of ionic bond is stronger than the attraction of polar water molecules
Giant molecular structures: Silicon oxide is insoluble in water
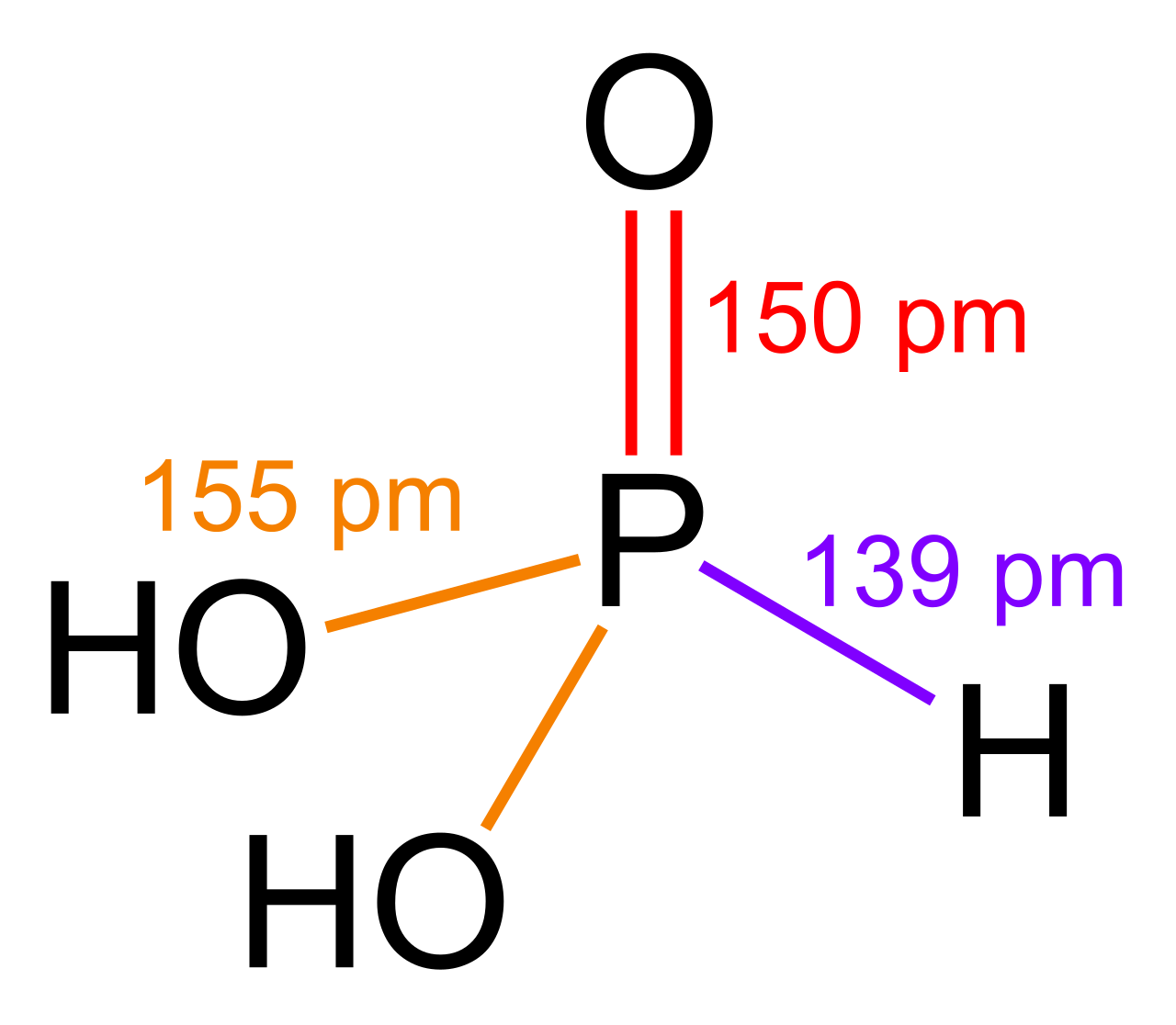
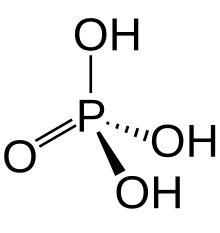
Small covalently-bonded molecules (all soluble): Phosphorus(III) oxide, \[\ce{P4O6 + 6 H2O -> 4 H3PO3}\], phosphorous acid; phosphorus(V) oxide, \[\ce{P4O10 + 6 H2O -> 4 H3PO4}\], phosphoric acid; sulfur dioxide, \[\ce{SO2 + H2O -> H2SO3}\]; and sulfur trioxide, \[\ce{SO3 + H2O -> H2SO4}\]
Acid/Base behaviour of oxides
The trend in acid-base behaviour is shown in various reactions, but as a simple summary: The trend is from strongly basic oxides on the left-hand side (\[\ce{Na}\]) to strongly acidic ones on the right (\[\ce{Cl}\]), with an amphoteric oxide (aluminium oxide, which shows both acidic and basic properties) in the middle. For this simple trend to hold, you have to be looking only at the highest oxides of the individual elements.
Basic oxides (react with water to form a base, or with an acid to form a salt and water in a neutralization reaction): \[\ce{Na2O + 2 HCl -> NaCl + H2O}\] and \[\ce{MgO + H2SO4 -> MgSO4 + H2O}\]
Amphoteric oxides (react with both acids and bases): Aluminium oxide: Aluminium and oxygen have a difference in electronegativity of 2.0, it exhibits both ionic and covalent nature as some of the electrons donated to oxygen are attracted towards the aluminium ion, creating some electron density between the aluminium and oxygen ions (which is a characteristic of covalent bonding). The reason for the ability to be both a base and acid.
Base: \[\ce{Al2O3 + 6 HCl -> 2 AlCl3 + 3 H2O}\]
Acid: \[\ce{Al2O3 + 2 NaOH + 3 H2O -> 2 NaAl(OH)4}\]
Acidic oxides:
Phosphorus(III) oxide, \[\ce{P4O6 + 8 NaOH -> 4 Na2HPO3 + 2 H2O}\], sodium phosphite;
phosphorus(V) oxide, \[\ce{P4O10 + 12 NaOH -> 4 Na3PO4 + 6 H2O}\], sodium phosphate;
sulfur dioxide, \[\ce{SO2(g) + 2 NaOH(aq) -> Na2SO3(aq) + H2O(l)}\] then \[\ce{Na2SO3(aq) + SO2(g) + H2O(l) -> 2 NaHSO3(aq)}\];
sulfur trioxide, \[\ce{SO3 + 2 NaOH -> Na2SO4 + H2O}\]
Acid/Base hebaviour of hydroxides
Sodium hydroxide: \[\ce{2 NaOH + H2SO4 -> Na2SO4 + 2 H2O}\], sodium sulfate
Magnesium hydroxide: \[\ce{Mg(OH)2(aq) + H2SO4(aq) -> MgSO4(aq) + 2 H2O(l)}\]
Aluminium hydroxide:
As base: \[\ce{2 Al(OH)3(s) + 3 H2SO4(aq) -> Al2SO4(aq) + 6 H2O(l)}\]
As acid: \[\ce{Al(OH)3(s) + NaOH(aq) -> NaAl(OH)4(aq)}\]
Chlorides with water
\[\ce{NaCl}\], \[\ce{MgCl2}\] and \[\ce{AlCl3}\]: dissolves in water to become hydrated
\[\ce{SiCl4}\] and \[\ce{PCl5}\]: reacts violently (hydrolysis) to form \[\ce{SiO2 + HCl}\] and \[\ce{H3PO4 + HCl}\]
Referenced by:
No backlinks found.