covalent bond
Covalent bonds
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
Types of formula representations

Single covalent bond
An example of a single covalent bond would be \[\ce{H-Cl}\], where one electron is shared from each atom. The bond here is usually a sigma bond.
Double covalent bond
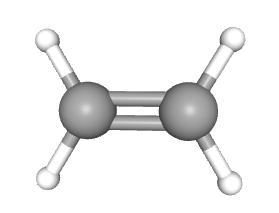
Ethene (\[\ce{C_{2}H_{4}}\]) has a double covalent bond, \[\ce{C=C}\], each carbon atom contributes two electrons to the covalent bond. The bonds here consists of one sigma bond and one pi bond.
Triple covalent bond
Similar to the bonds above, nitrogen gas has a triple covalent bond, \[\ce{N#N}\], where each nitrogen atom contributes three electrons. The bonds here consists of one sigma bond and two pi bonds.
Coordinate covalent bond
A coordinate covalent bond, sometimes known as dative bond is similar to covalent bonds, but instead both electrons that are shared comes from only one atom.

Formal charge
Sometimes atoms engage in covalent bonding by contributing more or less electrons than they have in their valence shell. The most common example is \[\ce{NH4+}\] where due to its dative covalent bond, it has an additional proton that is not matched by an electron. This net charge of +1 is referred to as a formal charge.
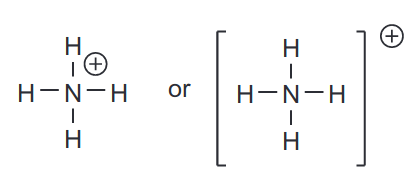
Another example would be a species known as carbanions, which carries a net charge of -1.
Energy changes in the formation of covalent bonds
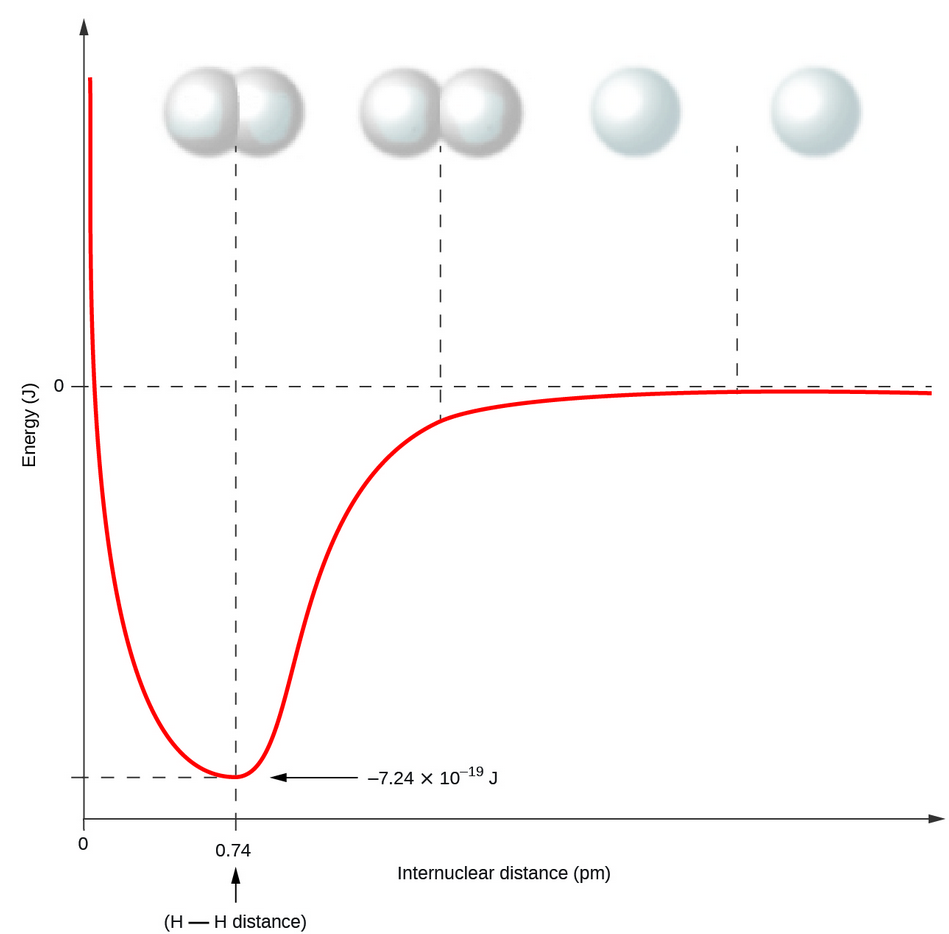
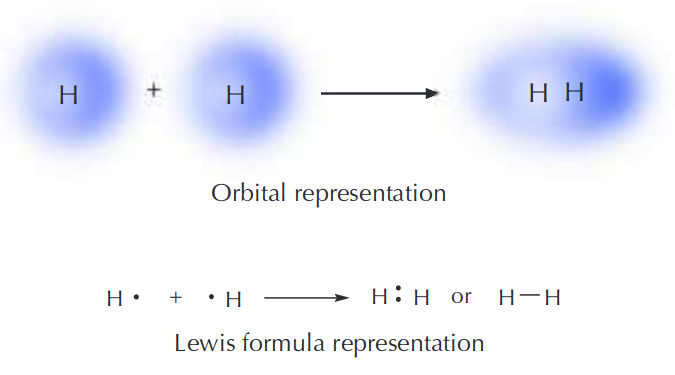
For instance, the hydrogen molecule, \[\ce{H2}\] contains a single covalent bond between its two hydrogen atoms. As they approach each other, their valence orbital (1s) begin to overlap which decreases their energy. Mathematically, the reason why overlap is good is because it signifies there is constructive interference of the wave functions. This translates to a higher probability of finding an electron in that area/electron effects are felt more there. The reason for this is because at the point way they overlap, classically we would say the conditions are more 'favourable' for the electron: there is a greater force from nuclei, less repulsion from other electrons, etc. We can describe this 'favoured' state, which is less likely to change to an unfavourable state, as a 'lower energy' or 'more stable' state. So, essentially stronger overlap = more electron density = stronger bond.
Difference between ionic bonds
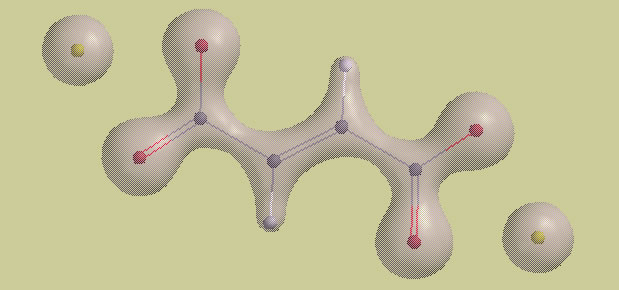
Covalent bonds are associated with a build-up of electron density between the bonded atoms. Ionic bonds do not involve electron sharing, and do not show this build-up. The following salt, disodium fumarate, \[\ce{Na2C4H2O4}\], is held together by a combination of covalent and ionic bonds, so its electron density cloud should reveal both of these characteristic patterns.
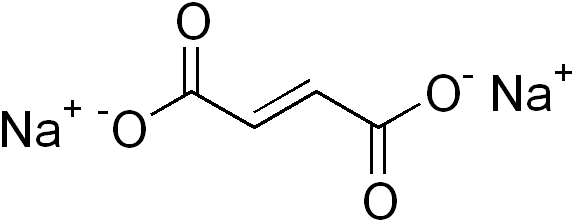
Electron density of disodium fumarate: