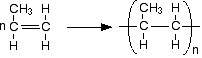alkene
Alkene
Reactions
Dehydrohalogenation of haloalkane
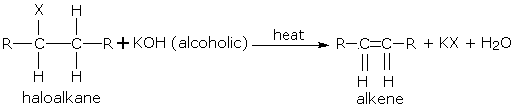
Haloalkane added with ethanolic \[\ce{NaOH}\] (\[\ce{NaOH}\] dissolved in ethanol) when heated produces alkene.
Dehydration of alcohol
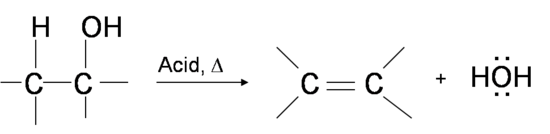
The reagents to this reaction is either concentrated acid such as \[\ce{H2SO4}\] or a heated catalyst such as \[\ce{Al2O3}\].
Oxidation of alkene with hot concentrated acidified \[\ce{KMnO4}\]
The acidified \[\ce{KMnO4}\] solution oxidizes the alkene by breaking the carbon-carbon double bond and replacing it with two carbon-oxygen double bonds.
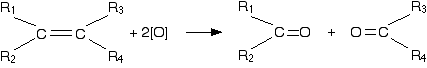
The products are known as carbonyl compounds because they contain the carbonyl group, \[\ce{C=O}\].
Now, here's where the reaction branches out.
-
Both attached \[\ce{R}\] groups are alkyl (\[\ce{-CH3}\]) groups
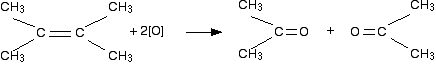
These carbonyl compounds are now identified as ketones. Ketones aren't that easy to oxidize, and so there is no further action. Though is a gross over-simplification. In practice, ketones are also oxidized by \[\ce{KMnO4}\] under these conditions. The reaction is untidy and results in breaking carbon-carbon bonds either side of the carbonyl group. This is because \[\ce{KMnO4}\] is a devastating oxidizing agent, which is also why it is rarely used in organic chemistry reactions.
-
One hydrocarbon group and one hydrogen
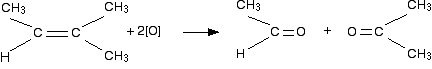
In this case, the products we get are an aldehyde and a ketone. As mentioned above, it's simpler to assume ketone doesn't oxidise anymore. However, aldehydes do oxidize further to give carboxylic acid.
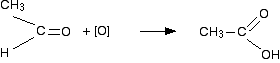
-
Two hydrogens, but no hydrocarbon group
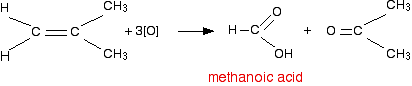
However, since methanoic acid is also easily oxidised by \[\ce{KMnO4}\], it will further get oxidised into carbon dioxide and water:
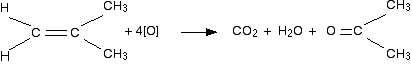
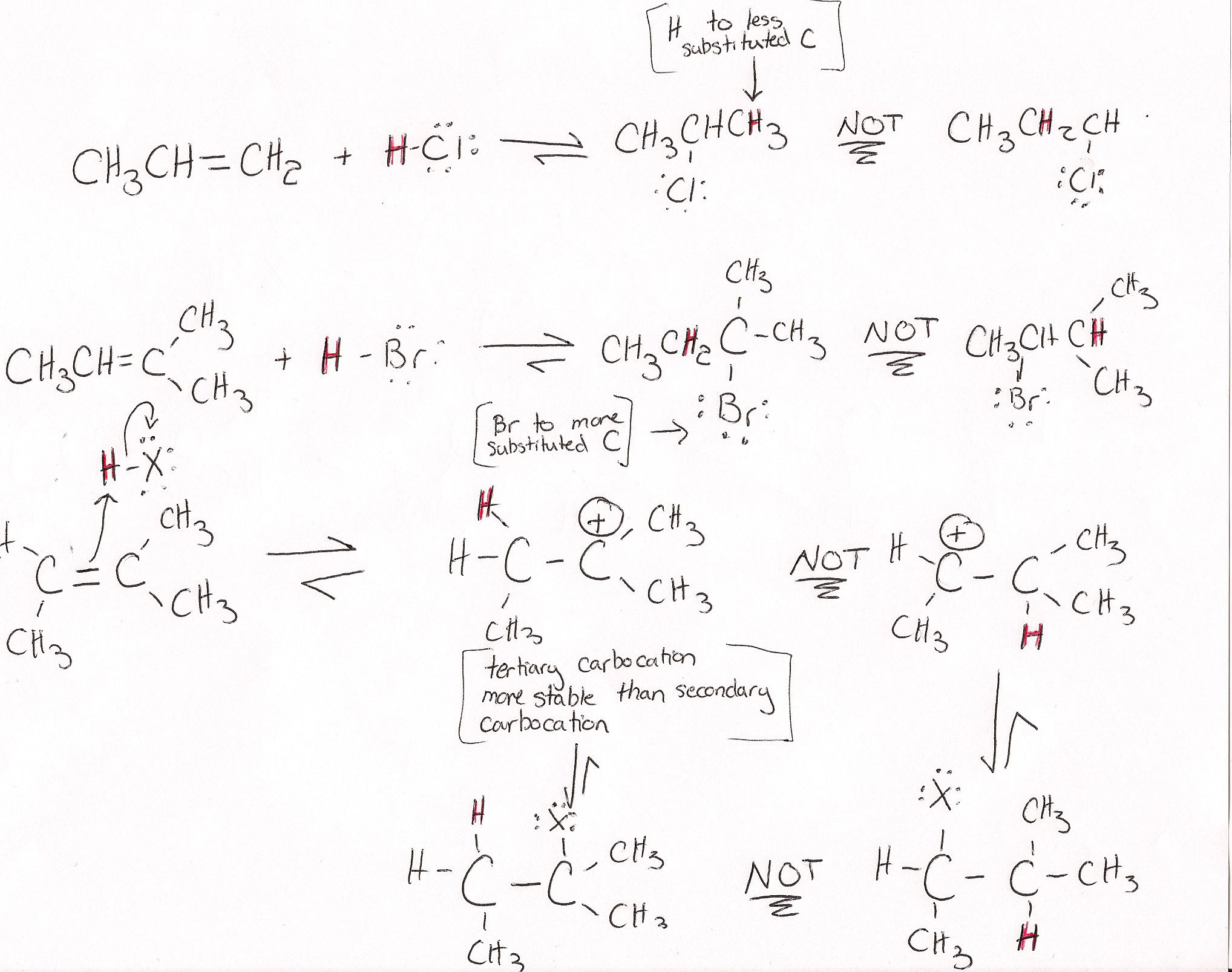
Electrophilic addition of hydrogen halides to alkene

\[\ce{\text{alkene} + HX ->[][\text{room temp.}] \text{halogenoalkane}}\]. This reaction follows Markovnikov's rule.
Electrophilic addition of halogens to alkene
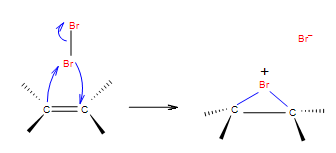
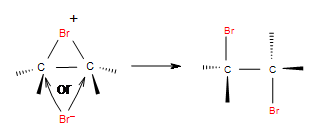
\[\ce{R2C=CR2 + X2 -> R2CX-CR2X}\].
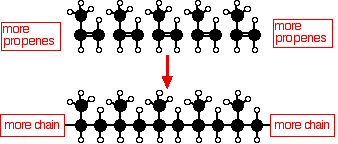
Addition polymerization of alkenes
Ethene is known as the monomer, while polythene is the polymer:
\[\ce{nCH2=CH2 -> [-CH2-CH2-]_{n}}\].
An addition reaction is one in which two or more molecules join together to give a single product. During the polymerisation of ethene, thousands of ethene molecules join together to make poly(ethene) - commonly called polythene.
Or, in the case of propene: