Bronsted-Lowry acid-base theory
Brønsted-Lowry acid-base theory
In the Arrhenius theory, acids are defined as substances that dissociate in aqueous solutions to give \[\ce{H+}\] (hydrogen ions or protons), while bases are defined as substances that dissociate in aqueous solutions to give \[\ce{OH-}\] (hydroxide ions). On the other hand, Brønsted-Lowry theory acids and bases are defined by the way they react with each other, generalising them.
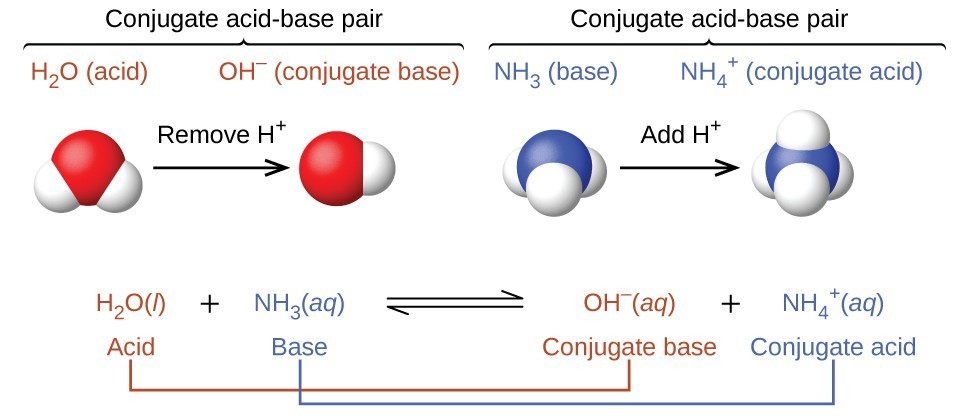
The equation can be written as: \[\ce{HA + B <=> A- + HB+}\], where the acid, \[\ce{HA}\], is a proton donor which can lose a proton to become its conjugate base, \[\ce{A-}\]. The base, \[\ce{B}\], is a proton acceptor which can become its conjugate acid, \[\ce{HB+}\]. Therefore, the proton donor is a Brønsted-Lowry acid, and the proton acceptor is the Brønsted-Lowry base.
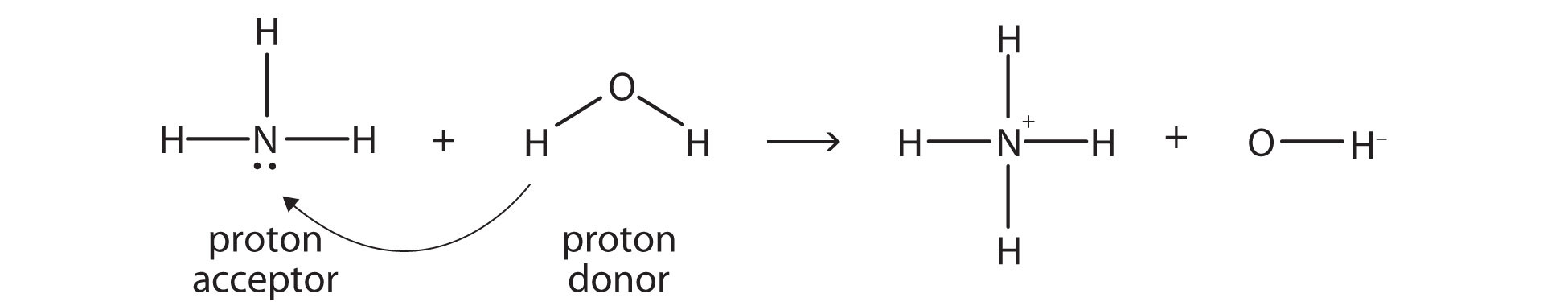
Ammonia (\[\ce{NH3}\]) is a base even though it does not contain \[\ce{OH-}\] ions in its formula. Instead, it generates \[\ce{OH-}\] ions as the product of a proton-transfer reaction with \[\ce{H2O}\] molecules; \[\ce{NH3}\] acts like a Brønsted-Lowry base, and \[\ce{H2O}\] acts like a Brønsted-Lowry acid.
Strength of acids
The strength of an acid is determined by how well it disassociates in water, \[\ce{HA <=> H+ + A-}\], a strong acid has it's equilibrium lying far to the right, while a weak acid has it's equilibrium far to the left.
An acid dissociation constant, \[K_{a}\], is also used to quantitatively measure the strength of an acid in a solution. \[K_{a}\] is measured as \[\frac{[\ce{H+}][\ce{A-}]}{[\ce{HA}]}\], where quantities in square brackets represent the molar concentrations (moldm-3) of the species at equilibrium. Usually, the form of \[\text{p}K_{a}\] is used because it provides a convenient logarithmic scale, where a lower \[\text{p}K_{a}\] would correspond to a stronger acid. \[\text{p}K_{a}\] in turn, is calculated with \[\text{p}K_{a}=-\log_{10}K_{a}\].