alcohol
Alcohol
Reactions
Hydration of alkenes with electrophilic addition
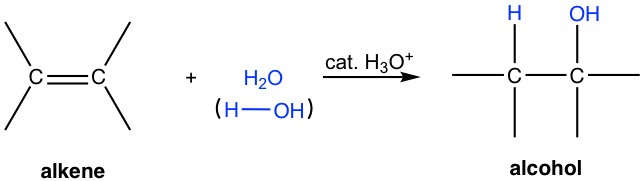
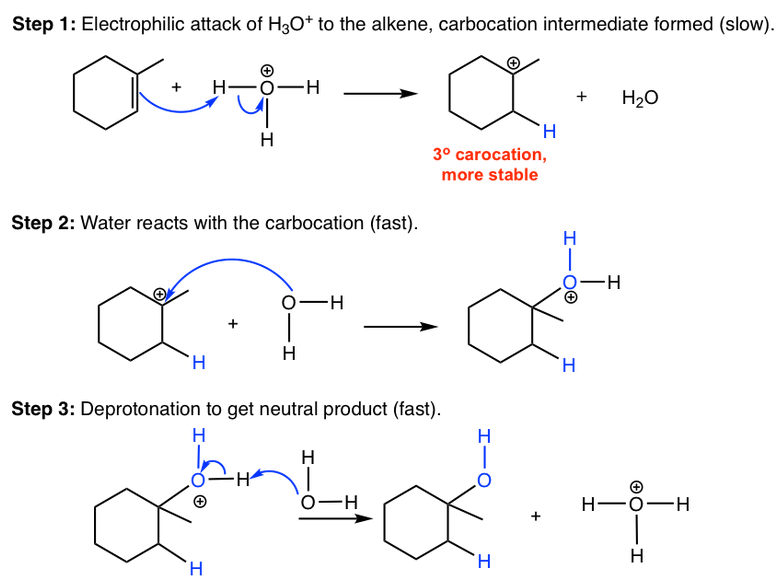
\[\ce{\text{alkene} + H2O(g) ->[\ce{H3PO4}] \text{alcohol}}\], where the acid is to catalyze this reaction. The phosphoric acid dissociates completely in aqueous solution and the \[\ce{H3O+}\] generated participates in the reaction.
Oxidation of alkenes to form -diol
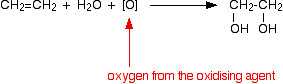
The reactant here is cold dilute acidified \[\ce{KMnO4}\] as the products of the reaction itself are also quite easily oxidised by manganate (VII) ions, which means that the reaction won't stop at this point unless the \[\ce{KMnO4}\] solution is very dilute and very cold.