electrophilic addition
Electrophilic addition
An electrophilic addition reaction is an addition reaction where a chemical compound containing a double or triple bond has a pi bond broken, with the formation of two new sigma bond. The driving force for this reaction is the formation of an electrophile \[\ce{X+}\] that forms a covalent bond with an electron-rich, unsaturated \[\ce{C=C}\] bond. The positive charge on \[\ce{X}\] is transferred to the carbon-carbon bond, forming a carbocation during the formation of the \[\ce{C-X}\] bond.
Mechanism
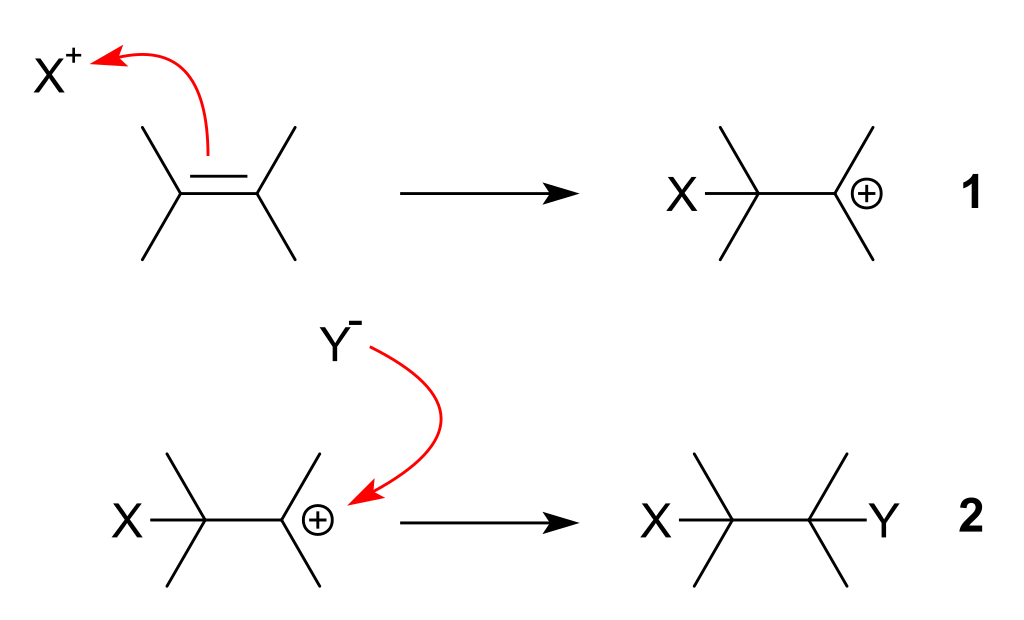
In the second step of an electrophilic addition, the positively charge on the intermediate combines with an electron-rich species to form the second covalent bond. The second step is the same nucleophilic attack process found in an SN1 reaction.
In step 1, the atomic orbitals look something like this:
As the electrophilic \[\ce{X}\] atom approaches the exposed pi bond, we much more likely to find the electrons in the half of the pi bond nearest to \[\ce{X-Y}\]. As the process continues, the two electrons in the pi bond move even further towards the \[\ce{X}\] until a covalent bond is made.
Then, in step 2, the electrons in the \[\ce{X-Y}\] bond are pushed entirely onto \[\ce{Y}\] to give a \[\ce{Y-}\] ion.
In the final stage of the reaction the electrons in the lone pair on the \[\ce{Y-}\] ion are strongly attracted towards the carbon. They move towards it and form a coordinate bond between \[\ce{Y}\] and the carbon.