free-radical substitution
Free-radical substitution
A radical-substitution reaction is a substitution reaction (a reaction in which one functional group in a chemical compound is replaced by another functional group) involving free radicals as a reactive intermediate. Similar to free-radical addition reactions, in it's initiation phase a free radical is created by homolysis.
Mechanism
The example used here would be the bromination of ethane, \[\ce{CH3CH3}\], to give bromomethane, \[\ce{CH3CH2Br}\]. \[\ce{CH3-CH3 + Br2 ->[$hv$] CH3-CH2-Br + H-Br}\], where \[hv\] represents visible light energy since the energy \[E\] of light is proportional to it's frequency \[v\], \[E=hv\] or more commonly known as Planck relation.
Initiation
Light energy breaks the \[\ce{Br-Br}\] bond giving two separate bromine radicals via homolytic cleavage, \[\ce{Br-Br ->[$hv$] 2Br^{.}}\].
or (for \[\ce{Cl2}\])
In fact, when we irradiate the reaction mixture with light, many \[\ce{Br2}\] molecules undergo homolysis and will more \[\ce{Br2}\] will continue to decompose as long as the mixture is irradiated with light. As a result, many \[\ce{Br^{.}}\] are produced at the same time thus starting many "chain" reactions.
Propagation
Each \[\ce{Br^{.}}\] formed has the ability to remove an \[\ce{H}\] from ethane, \[\ce{CH3-CH3 + Br^{.} -> CH3-CH2^{.} + H-Br}\]:
or more specifically,
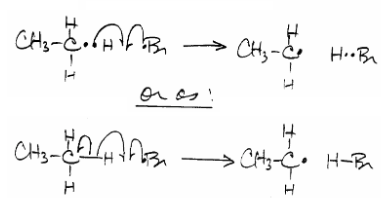
where either notation is acceptable (do note the single-barbed arrow).
Then due to the ethyl radical's high reactivity, \[\ce{CH3-CH2^{.} + Br-Br -> CH3-CH2-Br + Br^{.}}\]:
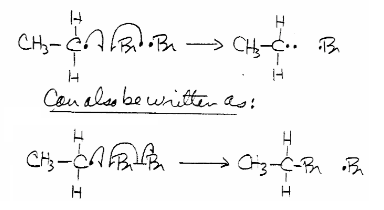
The \[\ce{Br^{.}}\] formed will react with another ethane molecule thus repeating this entire process again, or more specifically called a radical chain reaction.
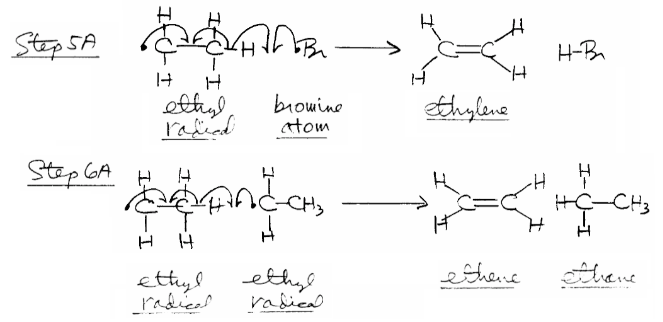
Termination
Since there are many radicals present at the same time, these radicals formed in initiation or in propagation reactions sometimes react with each other instead of reacting in the propagation steps. These reactions between two radicals lead to combination or disproportionation reactions that we collectively call termination reactions.
The \[\ce{Br^{.}}\] and \[\ce{CH3CH2^{.}}\] radicals can combine (a combination reaction) with each other in many different ways, such as through \[\ce{Br^{.} + Br^{.} -> Br2}\], \[\ce{CH3-CH2^{.} + Br^{.} -> CH3-CH2-Br}\] and \[\ce{CH3-CH2^{.} + CH3-CH2^{.} -> CH3-CH2-CH2-CH3}\] (forms butane, which happens very infrequently).
Besides undergoing combination reactions, two radicals sometimes react by disproportionation (a radical abstracts a hydrogen atom from an ethyl radical) by \[\ce{CH3-CH2^{.} + Br^{.} -> H-Br + CH2=CH2}\] and \[\ce{CH3-CH2^{.} + CH3-CH2^{.} -> CH3-CH3 + CH2=CH2}\].