sp3 hybridisation
\[\ce{sp^{3}}\] hybridisation
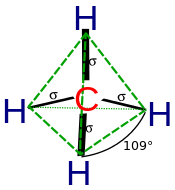
sp3 hybridization can explain the tetrahedral structure of molecules. In it, the 2s orbitals and all three of the 2p orbitals hybridise to form four \[\ce{sp^{3}}\] orbitals, each consisting of 75% p character and 25% s character. The frontal lobes align themselves in the manner shown below. In this structure, electron repulsion is minimized.
Energy changes:
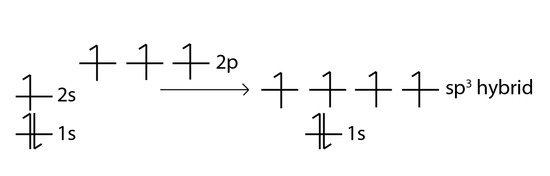
Hybridization of an s orbital with all three p orbitals (px, py, and pz) results in four \[\ce{sp^{3}}\] hybrid orbitals. \[\ce{sp^{3}}\] hybrid orbitals are oriented at bond angle of \[109.5^{\circ}\] from each other. This \[109.5^{\circ}\] arrangement gives tetrahedral geometry.
A methane molecule (\[\ce{CH_{4}}\]) is an example of a tetrahedrally coordinated carbon. The orbitals of carbon is initially:
This suggests that the carbon atom could use its two singly occupied p-type orbitals to form two covalent bonds with two hydrogen atoms in a methylene (\[\ce{CH_{2}}\]) molecule, with a hypothetical bond angle of \[90^{\circ}\] corresponding to the angle between two p orbitals on the same atom. However the true \[\ce{H-C-H}\] angle in singlet methylene is about \[102^{\circ}\] which implies the presence of some orbital hybridisation.
Much more energy is released in the formation of four bonds than two, so compounds of carbon with four bonds tend to be more stable than those with only two. Carbon does form compounds with only two covalent bonds (such as \[\ce{CH_{2}}\] or \[\ce{CF_{2}}\]), but these species are highly reactive, unstable intermediates that only form in certain chemical reactions.
The carbon atom can also bond to four hydrogen atoms in methane by an excitation of an electron via a small input of energy (externally, either from heat or light) from the doubly occupied 2s orbital to the empty 2p orbital, producing four singly occupied orbitals. The energy required is quite little as the energy gap between the 2s and 2p orbitals are small. This gives us a configuration of,
This carbon atom is now said to be in an excited state.
The electrons then rearrange themselves again in a process called hybridisation. This reorganises the electrons into four identical hybrid (accurately called sp3 hybrids) orbitals.
The energy released by the formation of two additional bonds more than compensates for the excitation energy required, thus energetically favoring the formation of four \[\ce{C-H}\] bonds.
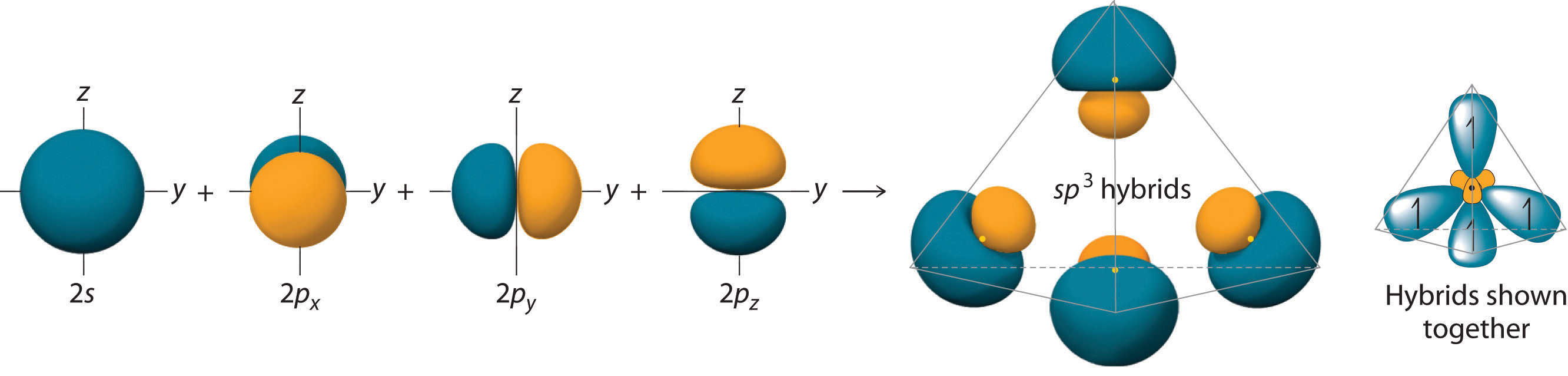
For longer alkane chains such as ethane, \[\ce{C2H6}\], the process is similar, each carbon atom promotes an electron then forms the hybrid orbitals.
However, here one of the sp3 hybrids from each carbon atom merges into a sigma bond.