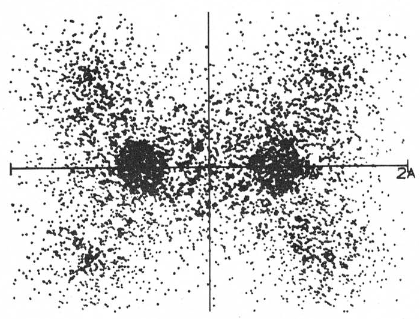
sigma bond
Sigma bond
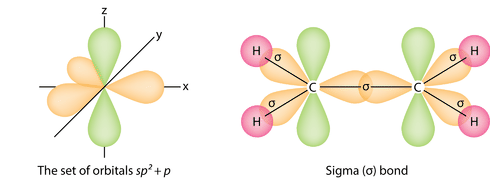
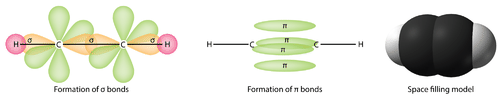
In chemistry, sigma bonds (\[\sigma\] bonds) are the strongest type of covalent bond. They are formed by head-on overlapping between atomic orbitals. Common forms of sigma bonds are \[s+s\], \[p_{z}+p_{z}\], \[s+p_{z}\] and \[d_{z^{2}}+d_{z^{2}}\] (see atomic orbitals for the shapes). Sigma bonds are the strongest type of covalent bonds due to the direct overlap of orbitals in an end-to-end fashion, with the electron density concentrated between the nuclei of the bonding atoms. The electrons in these bonds are sometimes referred to as sigma electrons.
Total electron density in the molecular plane of ethene (Moore, John W. and Davies, William G., 1976):