sp2 hybridisation
\[\ce{sp^{2}}\] hybridisation
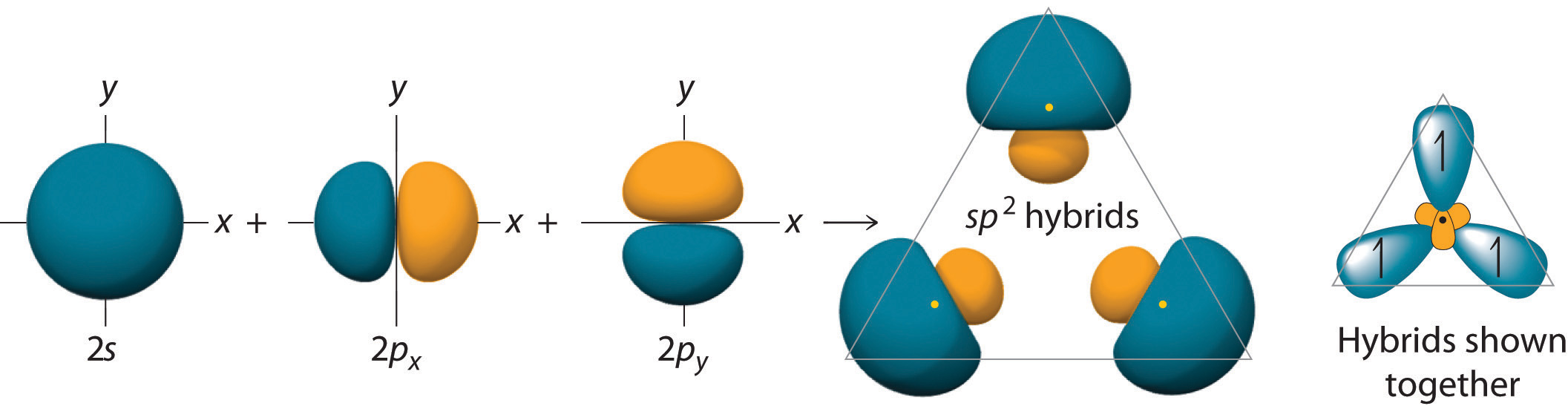
\[\ce{sp^{2}}\] hybridization can explain the trigonal planar structure of molecules. In it, the 2s orbitals and two of the 2p orbitals hybridize to form three sp orbitals, each consisting of 67% p and 33% s character. The frontal lobes align themselves in the trigonal planar structure, pointing to the corners of a triangle in order to minimize electron repulsion and to improve overlap. The remaining p orbital remains unchanged and is perpendicular to the plane of the three \[\ce{sp^{2}}\] orbitals.
Energy changes:
.jpg)
Hybridization of an s orbital with two p orbitals (px and py) results in three \[\ce{sp^{2}}\] hybrid orbitals that are oriented at \[120^{\circ}\] angle to each other. \[\ce{sp^{2}}\] hybridization results in trigonal geometry.
For example, ethene (\[\ce{C2H4}\]) has a double bond between the carbons. For ethene, carbon sp2 hybridises, unlike methane (as shown in sp3 hybridisation) because one pi bond is required for the double bond between the carbons and only three sigma bonds are formed per carbon atom. Initially, each carbon atom has the electron arrangement,
Now, in \[\ce{sp^{2}}\] hybridisation the 2s orbital is hybridised with only two of the three available 2p orbitals, more accurately the 2px and 2py orbitals. The third 2p orbital, 2pz remains unhybridised.
becomes,
A total of three sp2 orbitals with one remaining p orbital is formed. In ethene, the two carbon atoms form a sigma bond by overlapping one sp2 orbital from each carbon atom. The pi bond between the carbon atoms perpendicular to the molecular plane is formed by 2p-2p overlap. Each carbon atom forms covalent \[\ce{C-H}\] bonds with two hydrogens by \[\ce{s-sp^{2}}\] overlap, all with \[120^{{\circ}}\] bond angles. The \[\ce{C-H}\] bonds are all of equal strength and length.