electrophile
Electrophile
The concept of electrophilicity is relatively simple: an electron-poor atom is an attractive target for something that is electron-rich, i.e. usually a nucleophile. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through addition and substitution reactions.
The term related term electrophilic is an adjective that describes an atom, ion, or molecule that is electron-loving.
The electrophilic atom is quite commonly a carbon which is bonded to an electronegative atom, usually oxygen, nitrogen, sulfur, or a halogen.
Steric hindrance
Consider two hypothetical SN2 reactions, one in which the electrophile is a methyl carbon and another in which it is tertiary carbon.
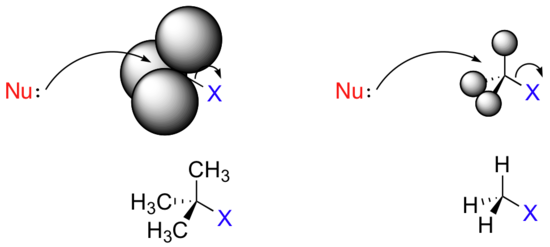
The three substituents on the methyl carbon electrophile are tiny hydrogen atoms, the nucleophile has a relatively clear path for backside attack. However, backside attack on the tertiary carbon is blocked by the bulkier methyl groups. Once again, steric hindrance caused by the bulky groups attached to the electrophile, rather than to the nucleophile, hinders the progress of an associative nucleophilic SN2 displacement.
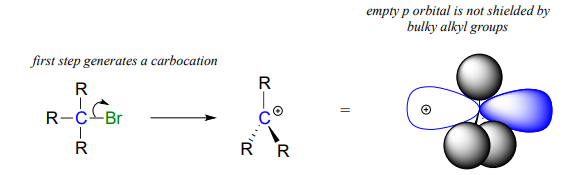

However, steric hindrance does not prevent a carbon from being a good electrophile, they only make it less likely to be attacked in a concerted (one-step) SN2 reaction. Nucleophilic substitution reactions in which the electrophilic carbon is sterically hindered are more likely to occur by a two-step, dissociative SN1 mechanism. This is because geometrically, the limitations imposed by sterics are significant mainly in an SN2 displacement, when the electrophile being attacked is a sp3-hybridised tetrahedral carbon with a relatively tight 109.4 degree angle. However in an SN1 mechanism, the leaving group leaves first, which allows the nucleophile to attack a sp2-hybridised carbocation intermediate, which has trigonal planar geometry with angles of 120 degrees.
Carbocation stability
An SN1 reaction requires a stabilized carbocation intermediate. The more stable the relevant carbocation intermediate, the more favored the SN1 reaction pathway. SN1 reactions in general do not occur at methyl or primary carbon electrophiles as the carbocation intermediates involved would be too unstable and the rate-determining carbocation generating step would have a very high energy barrier. Substitution on these electrophiles will likely occur through the SN2 pathway.
The SN1 reaction pathway is possible, however, with secondary and tertiary carbon electrophiles, or with any other carbon electrophile in which departure of the leaving group generates a carbocation which is stabilized by resonance.