enantiomer
Enantiomer
An enantiomer, also known as an optical isomer, is one of a pair of molecular entities which are mirror images of each other and non-superposable.
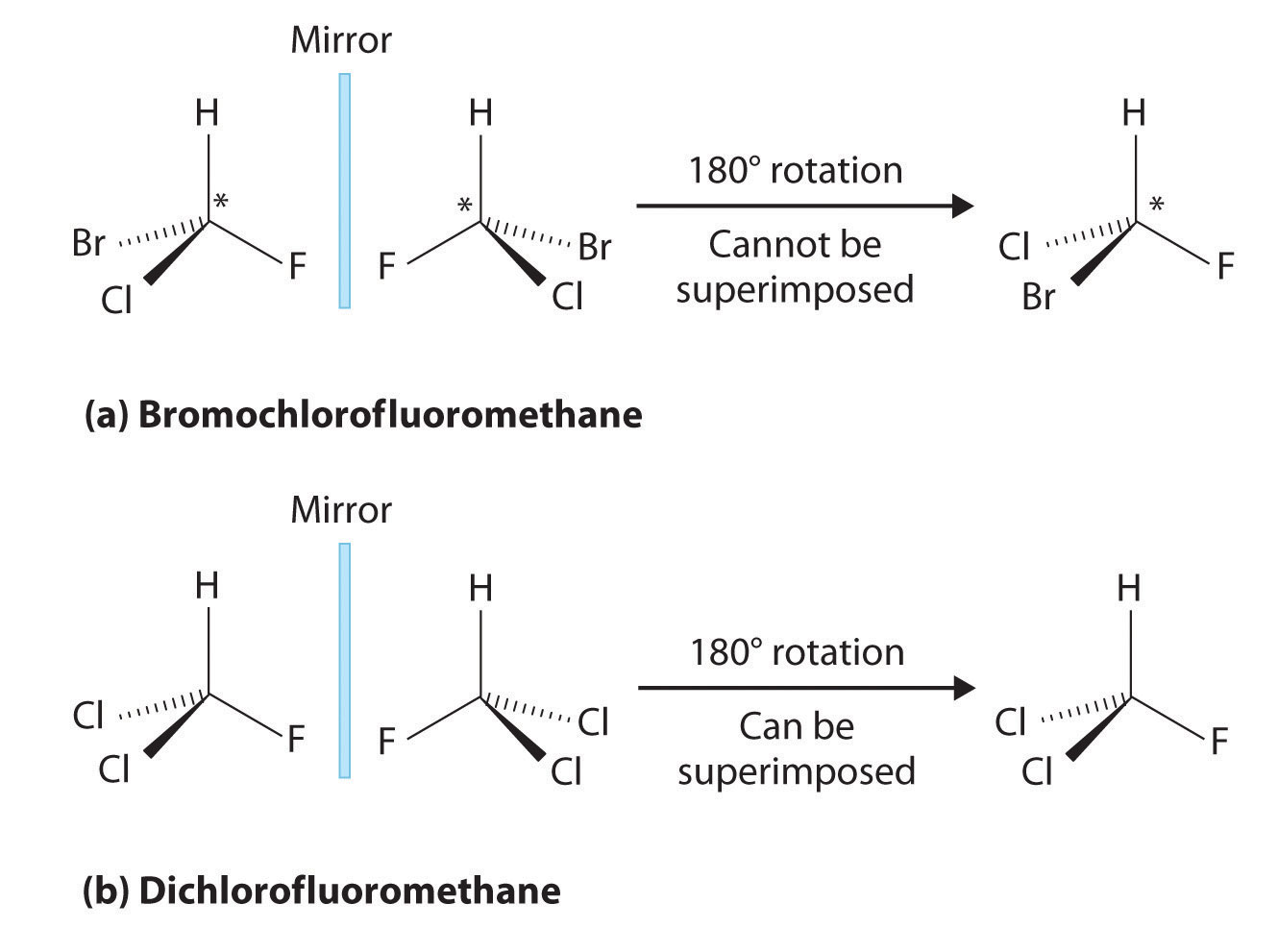
Here, we see that bromochlorofluoromethane is a chiral molecule whose stereocenter is designated with an asterisk while dichlorofluoromethane is not a chiral molecule.
Chirality
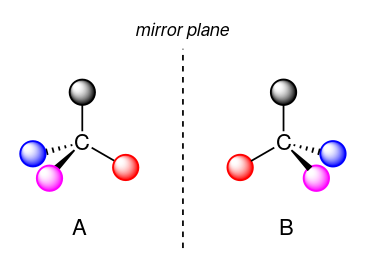
The term chiral, from the Greek work for "hand", refers to anything which cannot be superimposed on its own mirror image. Certain organic molecules are chiral meaning that they are not superimposable on their mirror image. Chiral molecules contain one or more chiral centers, which are almost always tetrahedral (sp3-hydridised) carbons with four different substituents.
Taking the example above as demonstration, if we were to flip A over,
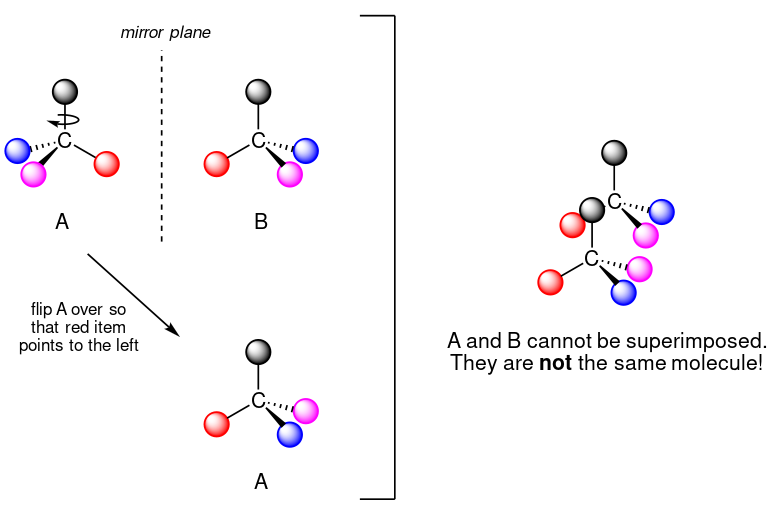
we will find that A is not superimposable on its mirror image B, thus by definition A is a chiral molecule. A and B are then called enantiomers/optical isomers.
Consider 2-butanol:
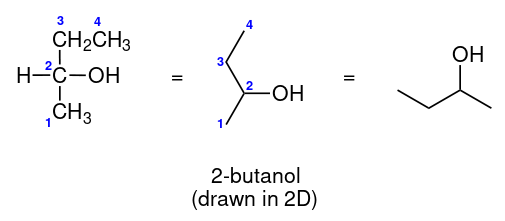
We can see that carbon number 2 is a chiral centre as it's sp3-hybridised, and the four substituents attached are a \[\ce{H}\], a methyl \[\ce{-CH3}\] group, an ethyl \[\ce{-CH2CH3}\] group and a hydroxyl \[\ce{OH}\] group.
If the bonding at C2 of 2-butanol is drawn in three dimensions:
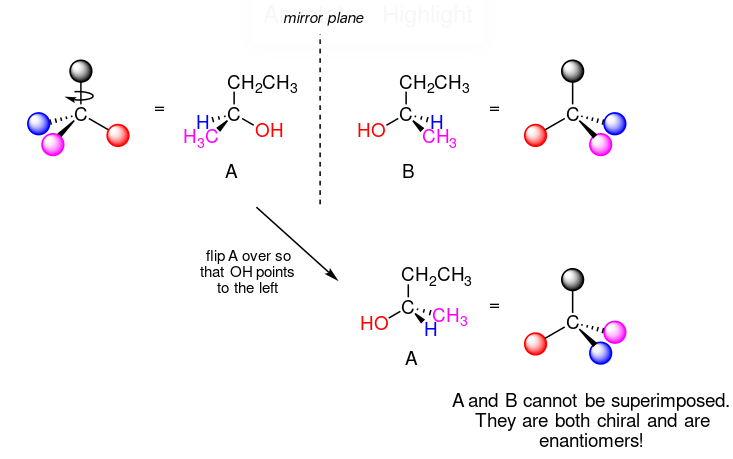
To confirm if it's chiral, when we try to superimpose A onto B, we find that it is impossible.
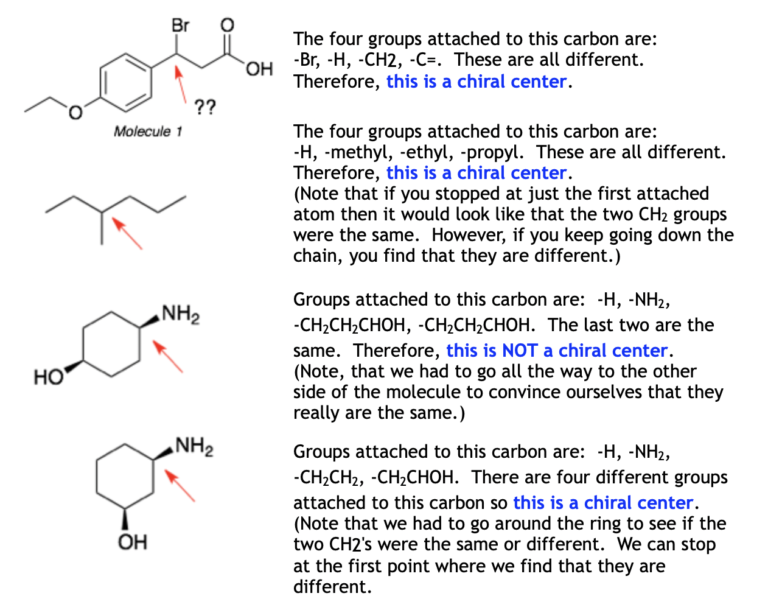
Finding chiral centres
- Eliminate the atoms that cannot be chiral centres. These include \[\ce{CH2}\] groups, \[\ce{CH3}\] groups, oxygen, halogen and any atom that is part of a double or triple bond.
- For the remaining atoms, list out the groups (substituents) attached to that atom. If there are four different groups, then it is a chiral centre.
Wedge and dash projection
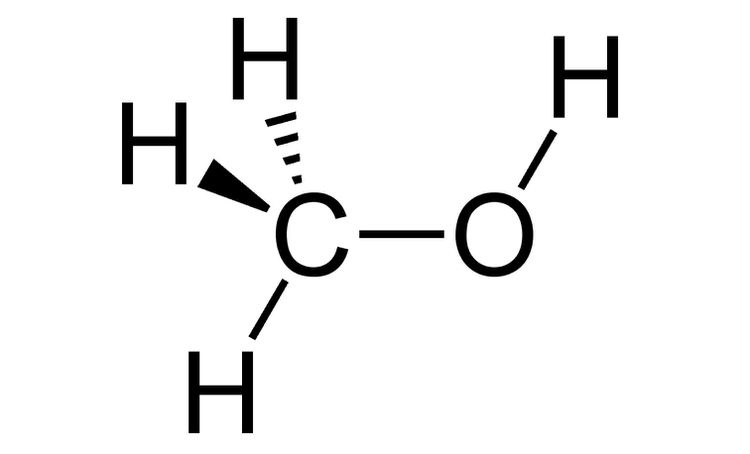
- Solid lines represent bonds that are in the plane of the paper
- Dashed lines to represent bonds that extend away from the viewer
- Wedge-shaped lines to represent bonds oriented facing the viewer
Referenced by:
No backlinks found.