orbital hybridisation
Orbital hybridisation
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds. It was introduced to explain molecular structures when the valence bond theory failed to correctly predict them.
Hybridization is a theoretical model used to describe the behavior of electrons in molecules, rather than an actual mechanism within atoms. It involves mathematical functions that approximate how orbitals combine to create molecular structures that closely resemble real-world observations. For instance, the base atomic orbitals in carbon, including a spherical s orbital and three p orbitals oriented along the x, y, and z axes at 90-degree angles, do not naturally explain the tetrahedral geometry of methane, which features bond angles of 109.5 degrees. To better represent this, parts of the s and p orbitals are mathematically combined to form new hybrid orbitals, termed \[\ce{sp^{3}}\], because they incorporate characteristics from one s and three p orbitals. These hybrid orbitals achieve orientations that allow for the observed bond angles in methane. While atoms are not aware of these theoretical constructs, the model effectively explains how electrons distribute themselves to minimize repulsion, given their negative charge, while maintaining attraction to the positive nuclei of carbon and hydrogen. This results in a structure that is described as \[\ce{sp^{3}}\] hybridized in methane, reflecting our mathematical approximation rather than the actual conditions within the atom.
\[\ce{sp^{3}}\] hybridisation
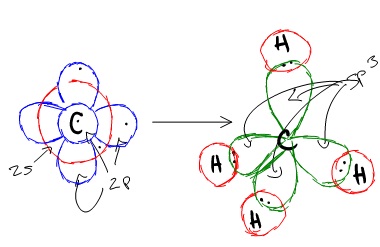
Carbon's ground state configuration is
According to the valence bond theory, carbon should form two covalent bonds resulting in \[\ce{CH2}\] because it has two unpaired electrons in its electronic configuration. However, experiments have shown that \[\ce{CH2}\] is highly reactive and cannot exist outside of a reaction. Therefore, this does not explain how \[\ce{CH4}\] can exist. To form four bonds the configuration of carbon must have four unpaired electrons. One way the formation of \[\ce{CH4}\] can be explained is that the 2s and the 3 2p orbitals combined to make four equal energy \[\text{sp$^{3}$}\] hybrid orbitals, giving us the configuration,
\[\ce{sp^{2}}\] hybridisation
\[\ce{sp}\] hybridisation
\[\text{sp}^{x}\text{d}^{y}\] hybridisation
Molecule shapes
Hybridisation helps to explain molecule shape, since the angles between bonds are approximately equal to the angles between hybrid orbitals. This is in contrast to VSEPR theory, which can be used to predict molecular geometry based on empirical rules rather than on valence-bond or orbital theories.
Determining sp/sp2/sp3 hybridisation
One can easily determine the hybridisation of an atom by looking at its steric number (as explained in VSEPR theory). If the steric number of an atom is 2, it corresponds to sp, 3 corresponds to sp2 and 4 corresponds to sp3.
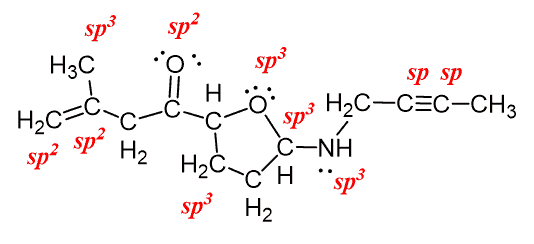
There are a number of exceptions such as for carbon dioxide, \[\ce{O=C=O}\], despite the carbon having two double bonds, it's sp hybridised as the optimal geometry is to keep both \[\ce{O}\] atoms at 180 degrees. The other two 2p orbitals are used for making the double bonds on each side of the carbon.