energy level
Energy level
According to classical mechanics, energy levels are fixed distances where electrons may be found. It is currently not known why these electrons occupy fixed energy levels and not the space in between (as shown below).
Energy levels of different orbitals (also visualises the sequence of the filling of orbitals according to Aufbau principle):
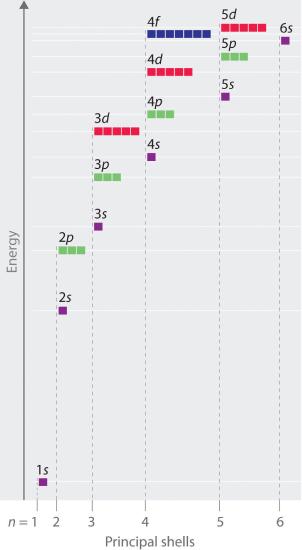
The higher the energy level occupied by an electron, the lesser energy it required to remove it from the nucleus. See effective nuclear charge for the reason.
Ground state
The ground state of an electron is the lowest energy state that it can occupy. It is the most stable state of an electron in an atom. In the ground state, an electron is in the lowest possible energy level, meaning that it cannot release energy and move to a lower level because no such lower level exists. The electron configuration in this state is such that the electrons fill the orbitals starting from the lowest energy level up, according to the Pauli Exclusion principle and the Aufbau principle.
Ground state does not necessarily correspond to being at the 1s orbital, but rather when all the electrons of an atom are in the lowest possible energy states (meaning that the energy levels have been filled in order of increasing energy).
Excited state
An excited state is any state of an electron that has a higher energy than the ground state. When an electron is in an excited state, it means it has absorbed energy (e.g., from light or heat) and has moved to a higher energy orbital. This state is less stable compared to the ground state, and the electron tends to return to the ground state by releasing energy, typically in the form of electromagnetic radiation (photons).
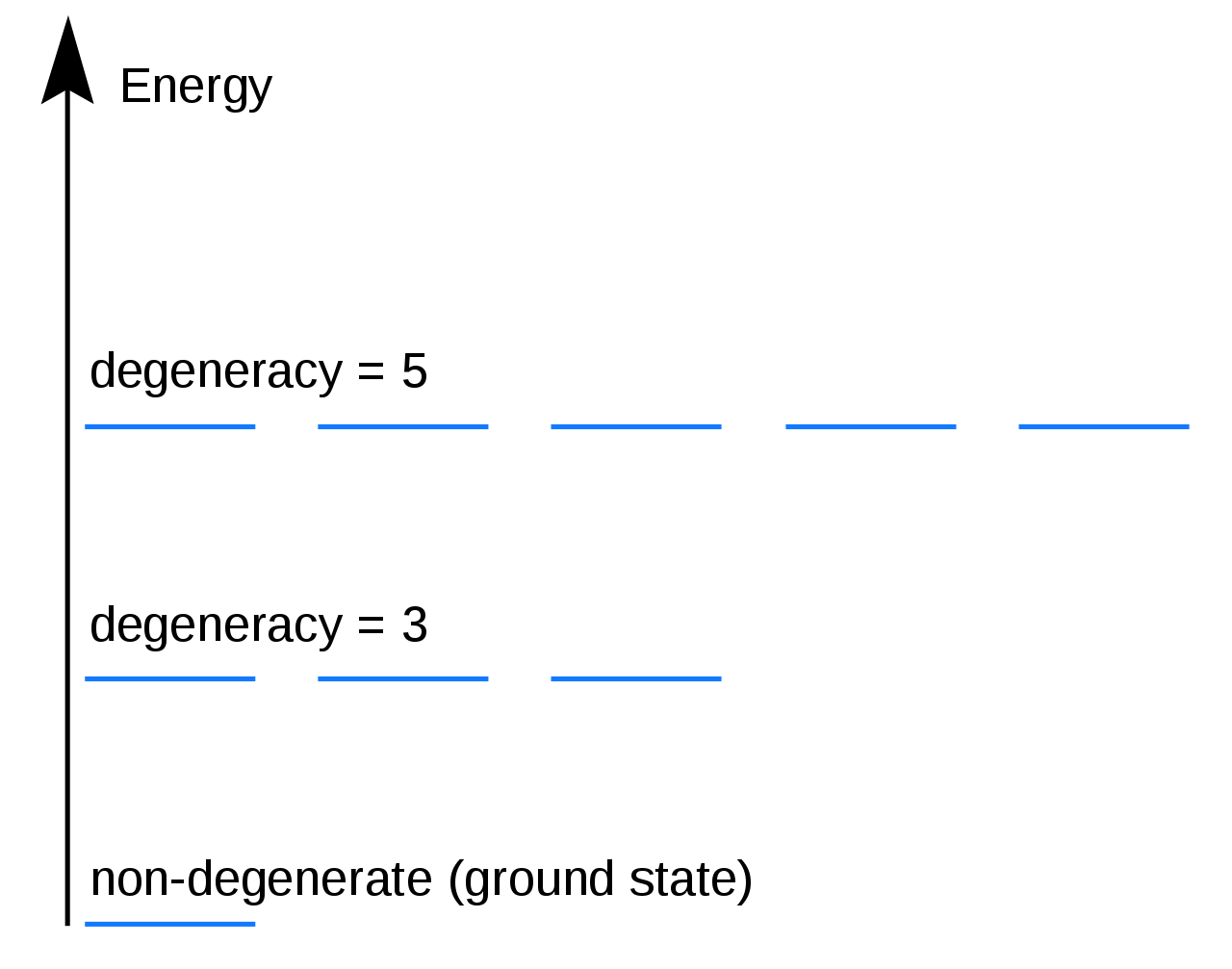
Degenerate energy levels
An energy level is degenerate if it corresponds to two or more different measurable states (states with different quantum numbers) of a quantum system. Conversely, two or more different states of a quantum mechanical system are said to be degenerate if they give the same value of energy upon measurement. For example, the three 2p orbitals are degenerate, meaning they have the same energy.
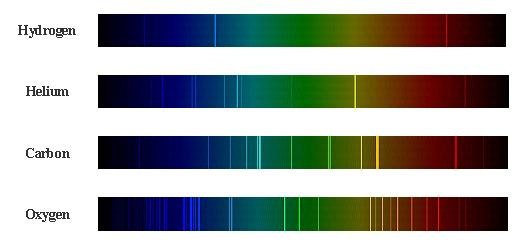
Energy levels in different kinds of atoms
Each different kind of atom, like hydrogen or radon, has a distinct nuclear charge and number of electrons. This makes the potential energy function different for each atom, and therefore results in different energy levels. This fact is reflected in the electromagnetic absorption or emission spectra of the atoms:
Energy levels in terms of quantum mechanics
According to quantum mechanics, electrons can be found anywhere inside the atom instead of a fixed energy level at any talking moment. But, there is a higher probability of finding them at these specific energy levels, again why this happens, nobody knows. If one try to understand it mathematically, fixed energy levels are because of the solution to Schodinger's Time-Independent equation tells so.