sp hybridisation
\[\ce{sp}\] hybridisation
sp hybridisation can explain the linear structure in molecules. In it, the 2s orbital and one of the 2p orbitals hybridise to form two sp orbitals, each consisting of 50% s and 50% p character. The front lobes face away from each other and form a straight line leaving a 180 degree angle between the two orbitals. This formation minimizes electron repulsion. Because only one p orbital was used, we are left with two unaltered 2p orbitals that the atom can use. These p orbitals are at right angles to one another and to the line formed by the two sp orbitals.
Energy changes:
.jpg)
These p orbitals come into play in compounds such as ethyne where they form two additional bonds, resulting in a triple bond. This only happens when two atoms, such as two carbons, both have two p orbitals each containing an electron. An sp hybrid orbital results when an s orbital is combined with p orbital, resulting in two sp hybrid orbitals since two orbitals (s and p each) were involved. sp hybridisation results in a pair of directional sp hybrid orbitals pointed in opposite directions. These hybridized orbitals result in higher electron density in the bonding region for a sigma bond toward the left of the atom and for another sigma bond toward the right. In addition, sp hybridization provides linear geometry with a bond angle of 180 degrees.
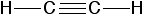
The chemical bonding in ethyne (\[\ce{C2H2}\]) consists of sp-sp overlap between the two carbon atoms forming a sigma bond and two additional pi bonds formed by p-p overlap. Each carbon also bonds to hydrogen in a pi bond s-sp overlap at \[180^{\circ}\] angles.
Similar to the other hybridisations, an electron from 2s is promoted to the empty 2pz orbital,
Since each carbon is only joining to two other atoms rather than four, only two of the orbitals are hybridised, forming two sp hybrids,
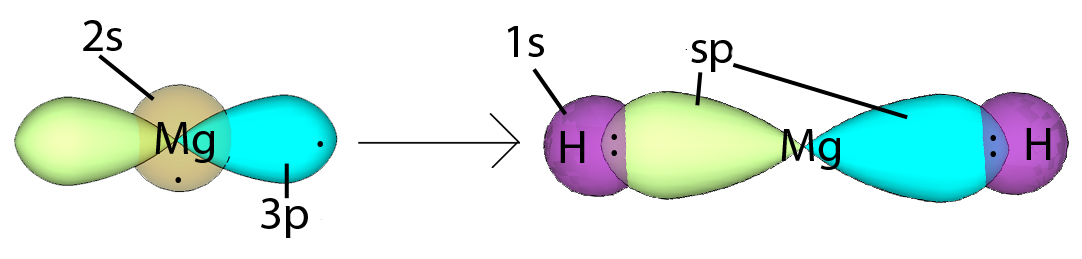
An illustration of how this looks like goes something like that,
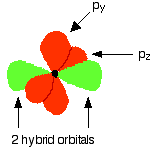
where each of the sp orbital form a sigma bond with the hydrogen or the adjacent carbon atom, while the remaining two orbitals from each carbon atom form pi bonds with each other to form a triple bond.
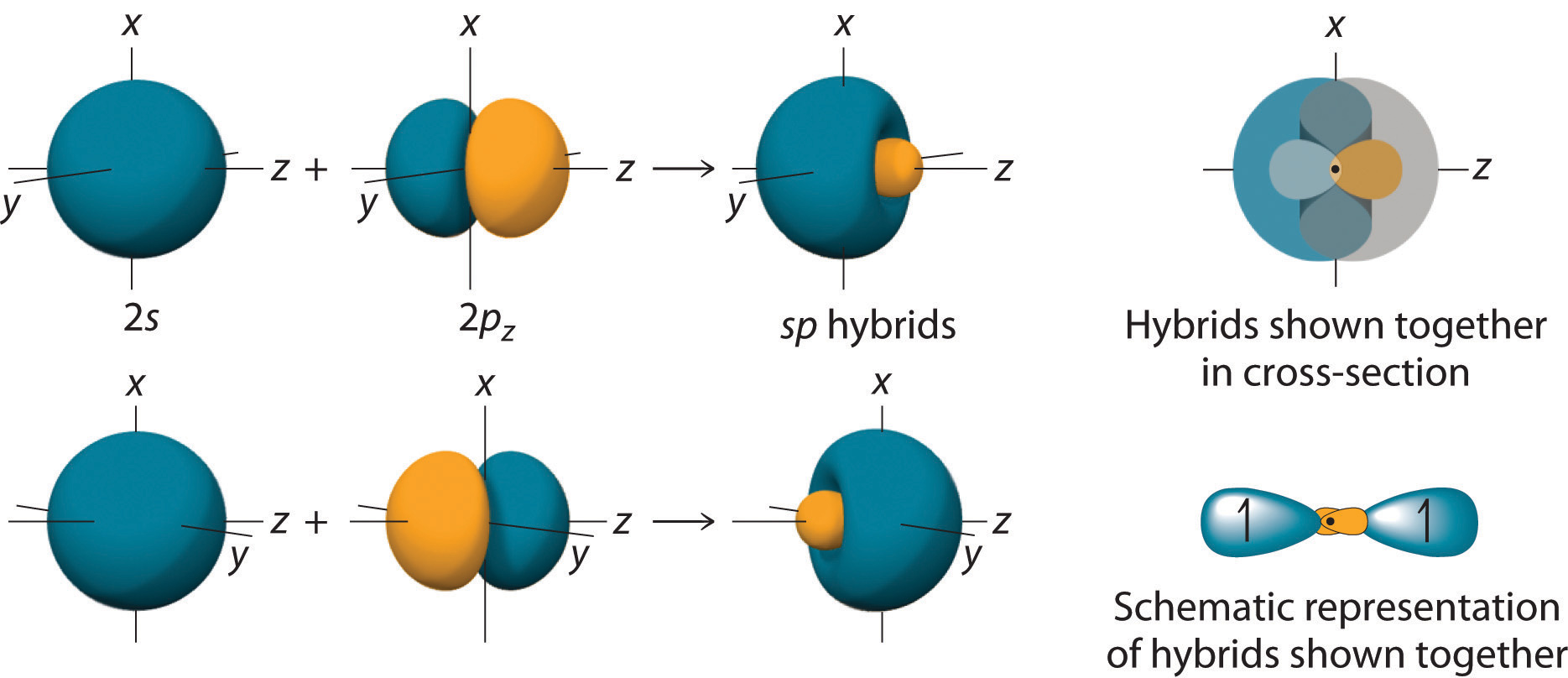
Magnesium hydride (\[\ce{MgH2}\]):