spxdy hybridisation
\[\text{sp}^{x}\text{d}^{y}\]
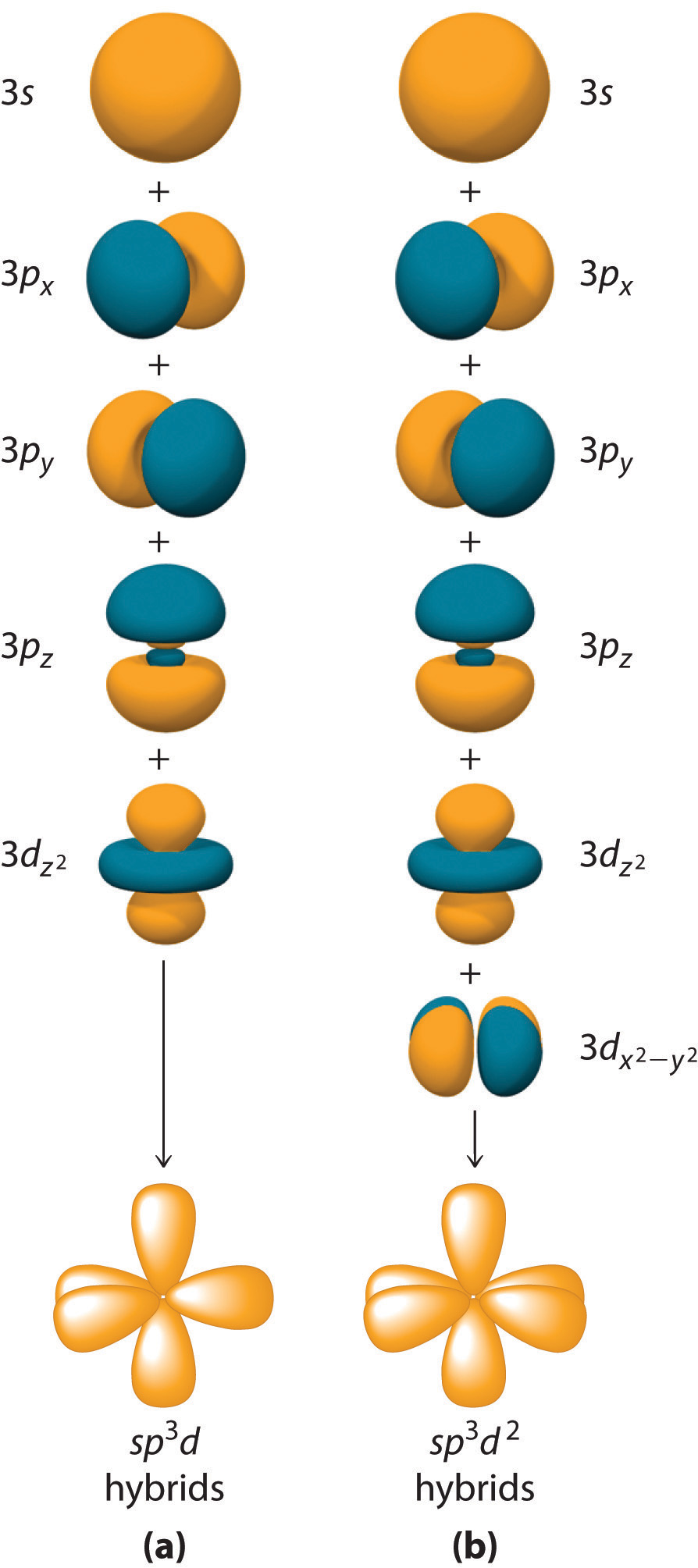
Hybridization is not restricted to the ns and np orbitals, where n is the principle quantum number. The bonding in compounds with central atoms in the period 3 and below can also be described using hybrid atomic orbitals. In these cases, the central atom can use its valence (n - 1)d orbitals as well as its ns and np orbitals to form hybrid atomic orbitals, which allows it to accommodate five or more bonded atoms (as in \[\ce{PF5}\] and \[\ce{SF6}\], see exceptions in octet rule). Using the ns orbital, all three np orbitals, and one (n - 1)d orbital gives a set of five \[\ce{sp^{3}d}\] hybrid orbitals that point toward the vertices of a trigonal bipyramid. In this case, the five hybrid orbitals are not all equivalent: three form a triangular array oriented at \[120^{\circ}\] angles, and the other two are oriented at \[90^{\circ}\] to the first three and at \[180^{\circ}\] to each other.
Similarly, the combination of the ns orbital, all three np orbitals, and two nd orbitals gives a set of six equivalent \[\ce{sp^{3}d^{2}}\] hybrid orbitals oriented toward the vertices of an octahedron. In the VSEPR theory, \[\ce{PF5}\] and \[\ce{SF6}\] are predicted to be trigonal bipyramidal and octahedral, respectively, which agrees with a valence bond description in which \[\ce{sp^{3}d}\] or \[\ce{sp^{3}d^{2}}\] hybrid orbitals are used for bonding.