electron shielding
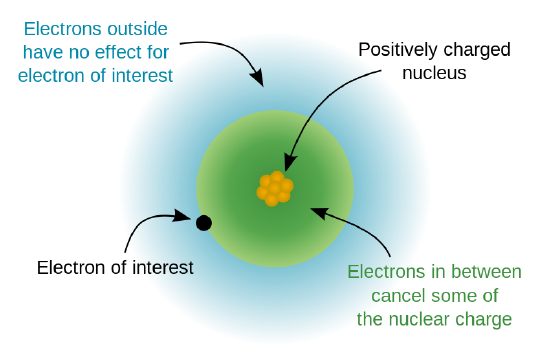
Electron shielding
If an electron is far from the nucleus of the atom, then at any given moment, many of the other electrons will be between that electron and the nucleus as illustrated below. Hence the electrons will cancel out a portion of the positive charge of the nucleus and thereby decreasing the attraction forces between the nucleus and the electron. As a result, the electron farther away will experience an effective nuclear charge that is less than the actual nuclear charge \[Z\].