electron subshells
Electron subshells
Each shell can be divided into multiple subshells labelled as s(harp), p(rincipal), d(iffuse), f(undamental).
| Shell | Number of subshells | Subshells | Maximum number of electrons |
| 1 | 1 | 1s | 2 |
| 2 | 2 | 2s, 2p | 8 |
| 3 | 3 | 3s, 3p, 3d | 18 |
| 4 | 4 | 4s, 4p, 4d, 4f | 32 |
So from the table above, we can see that:
| Subshell | Maximum number of electrons |
| s | 2 |
| p | 6 |
| d | 10 |
| f | 14 |
Each subshell is again, composed of atomic orbitals.
Filling of electron subshells
Electron filling always starts with 1s, the subshell closest to the nucleus. Next is 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, 8s, 5g as illustrated in the digram below. This is known as Aufbau principle. The filling of atomic orbitals however is governed by the Hund's rule of maximum multiplicity
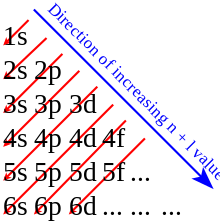
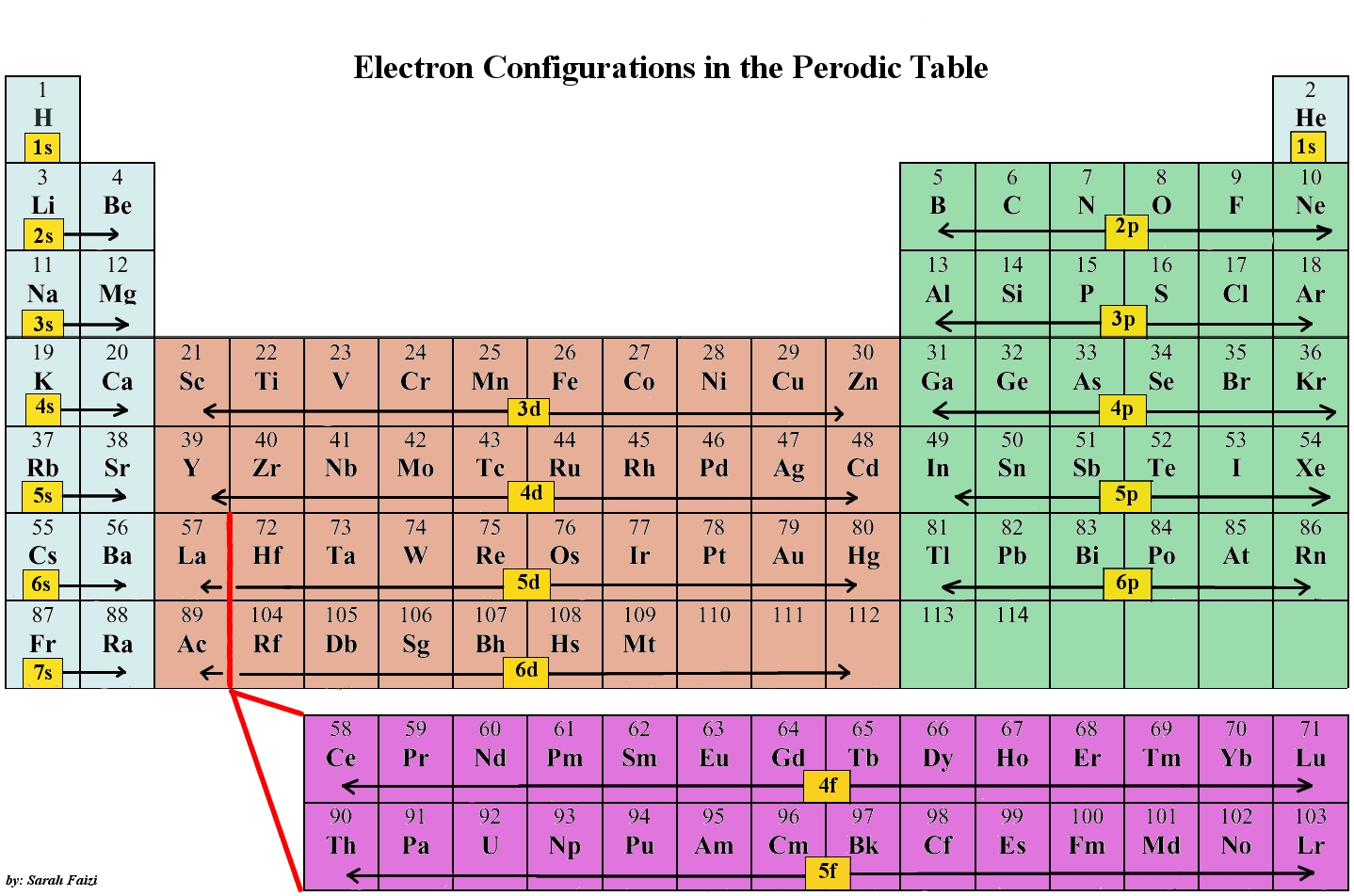
There are exceptions such as Cr and Cu, where it would be expected based on pattern that Cr would be [Ar] 3d44s2, instead it is [Ar] 3d54s1. There is no simple or rational explanation to this (yet). See section Exceptions in Aufbau principle.
The reason why the subshells fill up in this manner is due to effective nuclear charge.
Electrons in an orbital closer to the nucleus experience a higher \[Z_{\textit{eff}}\] because there is less electron shielding, and a high \[Z_{\textit{eff}}\] attraction lowers the potential energy of the electron. In nature, systems tend to move towards states of lower energy because these states are inherently more stable. Therefore, electrons will occupy the orbitals where they are most strongly attracted to the nucleus because these orbitals represent the lowest energy states.