ionisation energy
Ionisation energies
Ionisation energy is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, ion or molecule. The first ionisation energy is expressed as:
\[\ce{X(g) -> X+(g) + e-}\] which shows that the ionization is an endothermic process. Roughly speaking as per shown below, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy.
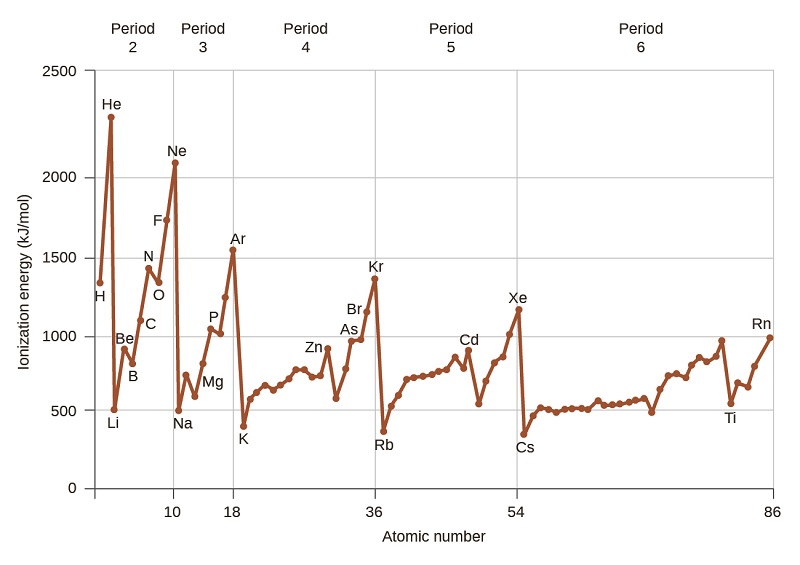
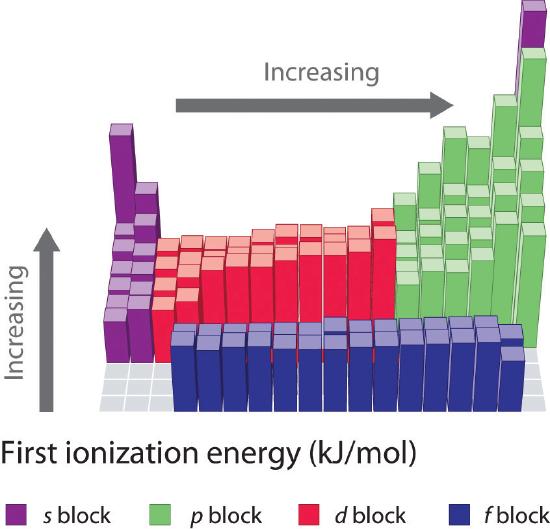
The ionization energy of atoms, denoted \[E_{i}\], is measured by finding the minimal energy of light quanta (photons) or electrons accelerated to a known energy that will kick out the least bound atomic electrons. The measurement is performed in the gas phase on single atoms. While only noble gases occur as monatomic gases, other gases can be split into single atoms.
Factors affecting ionisation energies in atoms
Nuclear charge
Increased number of protons will result in electrons getting held more tightly by the nucleus, therefore increasing ionisation energy.
Effective nuclear charge
Increased electron shielding and orbital penetration will cause electrons to be held less tightly by the nucleus and but will still result in increasing ionisation energies. While inner electrons do shield outer electrons from the full effect of the nuclear charge, it does not fully compensate for the increase in nuclear charge. This will lead to a tighter binding of the electrons, especially those in the outermost shell despite an increase in electron shielding.
Stability
An atom having a stable electron configuration.
Electron pairing energies
Half-filled subshells usually result in higher ionisation energies. Not always, see shortfalls of Aufbau principle.
Factors causing the decrease in ionisation energies
Transitioning across a new period
Alkali metals tend to easily lose electrons to form ions according to the octet rule as it is much more energetically favourable.
Moving from s-block to p-block or d-block to p-block
Due to the increased energy levels of consecutive orbitals (therefore the electron is, on average, to be found further from the nucleus) and shielding from the previous orbitals.
Occupying a p-subshell with its first electron with spin opposed to the other electrons
Such as in nitrogen (7N: 14.5 eV) to oxygen (8O: 13.6 eV), as well as phosphorus (15P: 10.48 eV) to sulfur (16S: 10.36 eV). The reason for this is because oxygen, sulfur and selenium all have dipping ionization energies because of electron pair repulsion. However, this discontinues starting from tellurium where the shielding is too small to produce a dip.
Factors causing the increase in ionisation energies
Approaching Group 18 noble gas elements
A complete electron subshell is most energetically favourable. It will require a large amount of energy to remove an electron from the atom.
Moving into d-block
The elements Sc with a 3d1 electronic configuration has a higher IP (21Sc: 6.56 eV) than the preceding element (20Ca: 6.11 eV), contrary to the decreases on moving into s-block and p-block elements. The 4s and 3d electrons have similar shielding ability: the 3d orbital forms part of the n=3 shell whose average position is closer to the nucleus than the 4s orbital and the n=4 shell, but electrons in s orbitals experience greater penetration into the nucleus than electrons in d orbitals. So the mutual shielding of 3d and 4s electrons is weak, and the effective nuclear charge acting on the ionized electron is relatively large. Yttrium (39Y) similarly has a higher ionisation energy (6.22 eV) than 38Sr: 5.69 eV.
Moving into f-block
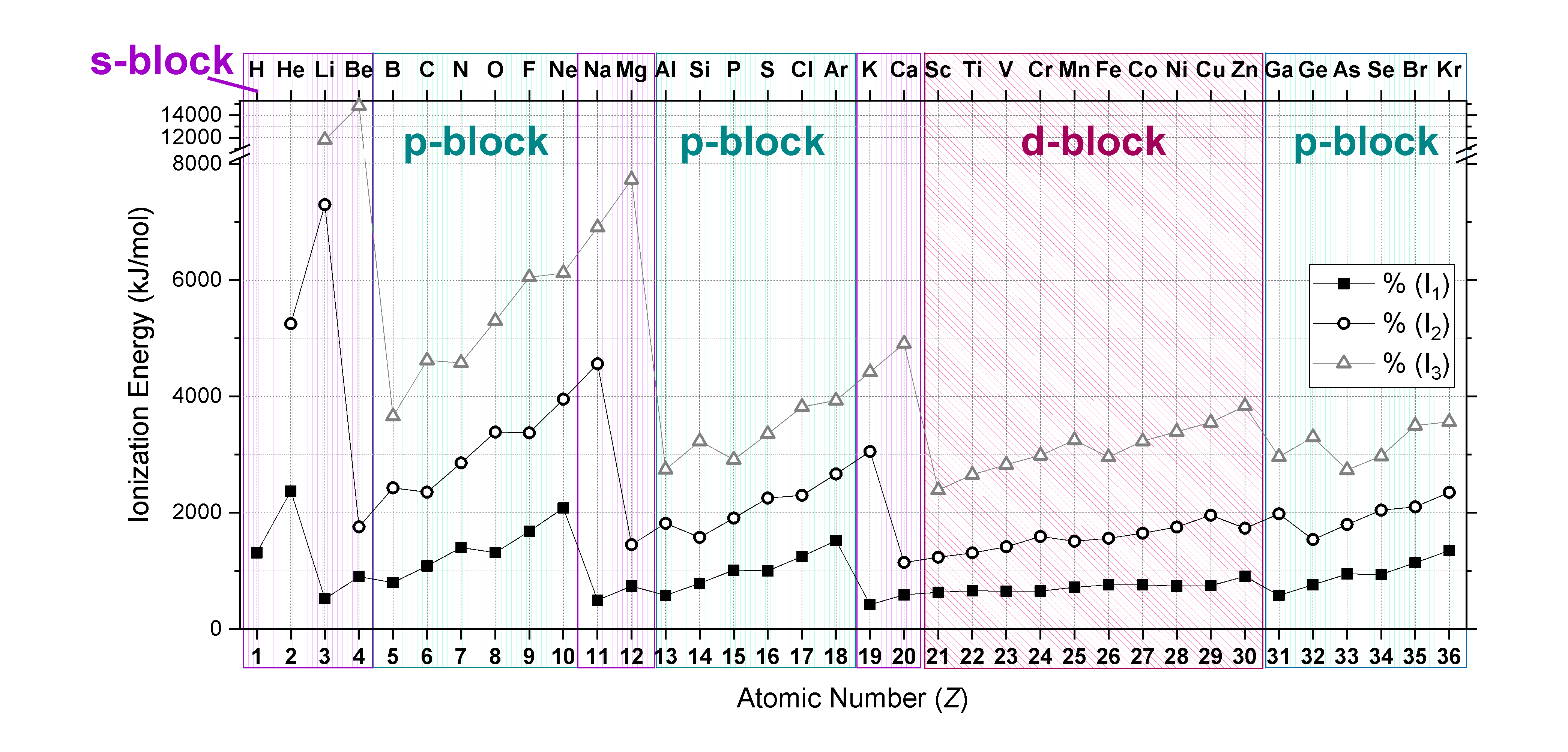
Successive ionisation energies
Successive ionization energies are generally much higher than the first due to changes in the atomic structure and electronic environment after each electron is removed. The consecutive ionisation energies can be written as: \[\ce{X^{+}(g) -> X^{2+}(g) + e-}\] and \[\ce{X^{2+}(g) -> X^{3+}(g) + e-}\], etc..
In a neutral atom, the outermost electrons are partially shielded from the full nuclear charge by the inner electrons. This shielding effect reduces the effective nuclear charge felt by the valence electrons, making them easier to remove. Once the first electron is removed, the remaining electrons experience a higher effective nuclear charge because there's one less electron to contribute to the shielding effect and the ratio of protons to electrons in the atom increases. This increased effective nuclear charge means that each successive electron feels a stronger pull from the nucleus.
Particularly notable increases in ionization energy occur when the removal of electrons starts to break into a closed-shell configuration, such as a noble gas configuration. These configurations are highly stable due to their full shells, and removing electrons from them requires significantly more energy.
Others