orbital penetration
Orbital penetration
Penetration describes the ability of an electron in a given subshell to penetrate within other shells and subshells to get close to the nucleus (the extent to which an electron can approach the nucleus). In a multi-electron system, electron penetration is defined by an electron's relative electron density (probability density) near the nucleus of an atom. Electrons in different orbitals have different wavefunctions and therefore different radial distributions and probabilities (defined by quantum numbers \[\ell\] and \[m_{\ell}\] around the nucleus). For example, we see that since a 2s electron has more electron density near the nucleus than a 2p electron, it is penetrating the nucleus of the atom more than the 2p electron.
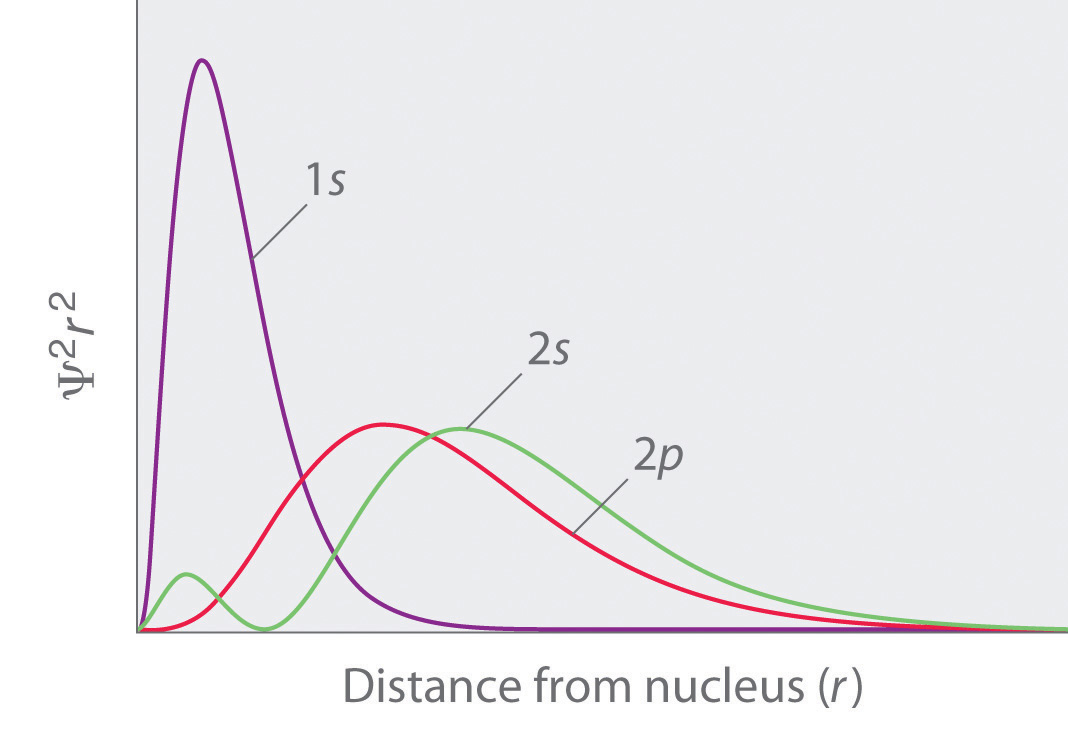
For different values of shell, \[n\] and subshell, \[\ell\], penetrating power of an electron follows this trend:
1s > 2s > 2p > 3s > 3p > 4s > 3d > 4p > 5s > 4d > 5p > 6s > 4f… (Aufbau principle)
Electrons which experience greater penetration experience stronger attraction to the nucleus, less shielding, and therefore experience a larger effective nuclear charge, \[Z_{\text{eff}}\], but shield other electrons more effectively.