ionic radius
Ionic radius
Defined as the radius of a monatomic (single) ion in an ionic crystal structure. The ions are ionic bonded to each other.
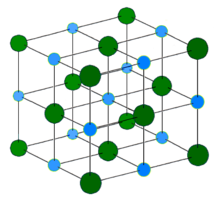
The diagram above shows the sodium chloride crystal structure.
Ionic radius of an ion may be larger or smaller than the neutral atom.
Larger
When an electron is added to the atom, forming an anion, the added electron increases the size of the electron cloud by interelectronic repulsion. Interelectronic repulsion refers to the force of repulsion between electrons in an atom due to their negative charges. According to Coulomb's law, like charges repel each other, so electrons in an atom naturally repel each other. As a result, the electrons spread out more to minimize the repulsive forces, leading to an expansion of the electron cloud and an increase in the size of the anion compared to the neutral atom.
Smaller
When an electron is removed from an atom, forming a cation, the number of electrons in the atom decreases while the number of protons in the nucleus remains the same. This results in a higher effective nuclear charge, which is the net positive charge that an electron experiences from the nucleus and the remaining electrons are pulled closer to the nucleus.