Coulomb's law
Coulomb's law
Coulomb's torsion balance
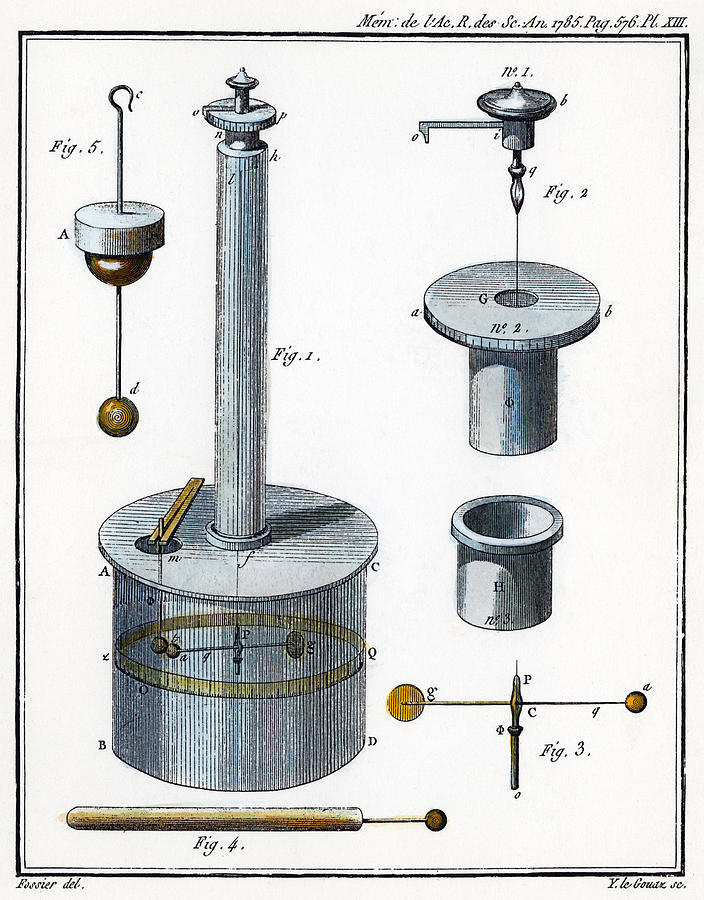
Source: Coulomb, C. A. (1788). Sur l'électricité et le magnétisme, premier mémoir. In. Mémoires de l'Academie Royale des Sciences pour l'année 1785, Paris, 576
Coulomb initially found that the magnitude of the electric force that a particle exerts on another is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. Mathematically speaking, \[F\propto \frac{q_{1}q_{2}}{r^{2}}\], where \[F\] refers to the magnitude of electrostatic force exerted on each particle. By Newton's third law, the force exerted on each particle is the same, just different direction, i.e. \[F_{21}=-F_{12}\]. It was also experimentally determined that the constant of proportionality, \[k_{e}\approx9.0\times10^{9}\text{ Nm$^{2}$C$^{-2}$}\].
Derivation of \[k_{e}\]
Originally Coulomb used the CGS-ESU unit system which gave him \[k_{e}=1\], as \[F\] was measured in dyne (\[10^{-5}\text{ N}\]), \[r\] measured in centimetres (\[10^{-2}\text{ m}\]) and \[q\] measured in statcoulombs (\[\approx3.3356\times10^{-10}\text{ C}\]) during his time. Now, to convert \[k_{e}\] into modern SI units,
which tallies with the value of \[k_{e}=8.98755\times10^{9}\text{ Nm$^{2}$C$^{-2}$}\] we use today (after more precise measurements).
Definition
Coulomb's law, is an experimental law of physics that calculates the amount of force between two electrically charged particles at rest. This electric force is conventionally called the electrostatic force or Coulomb force.
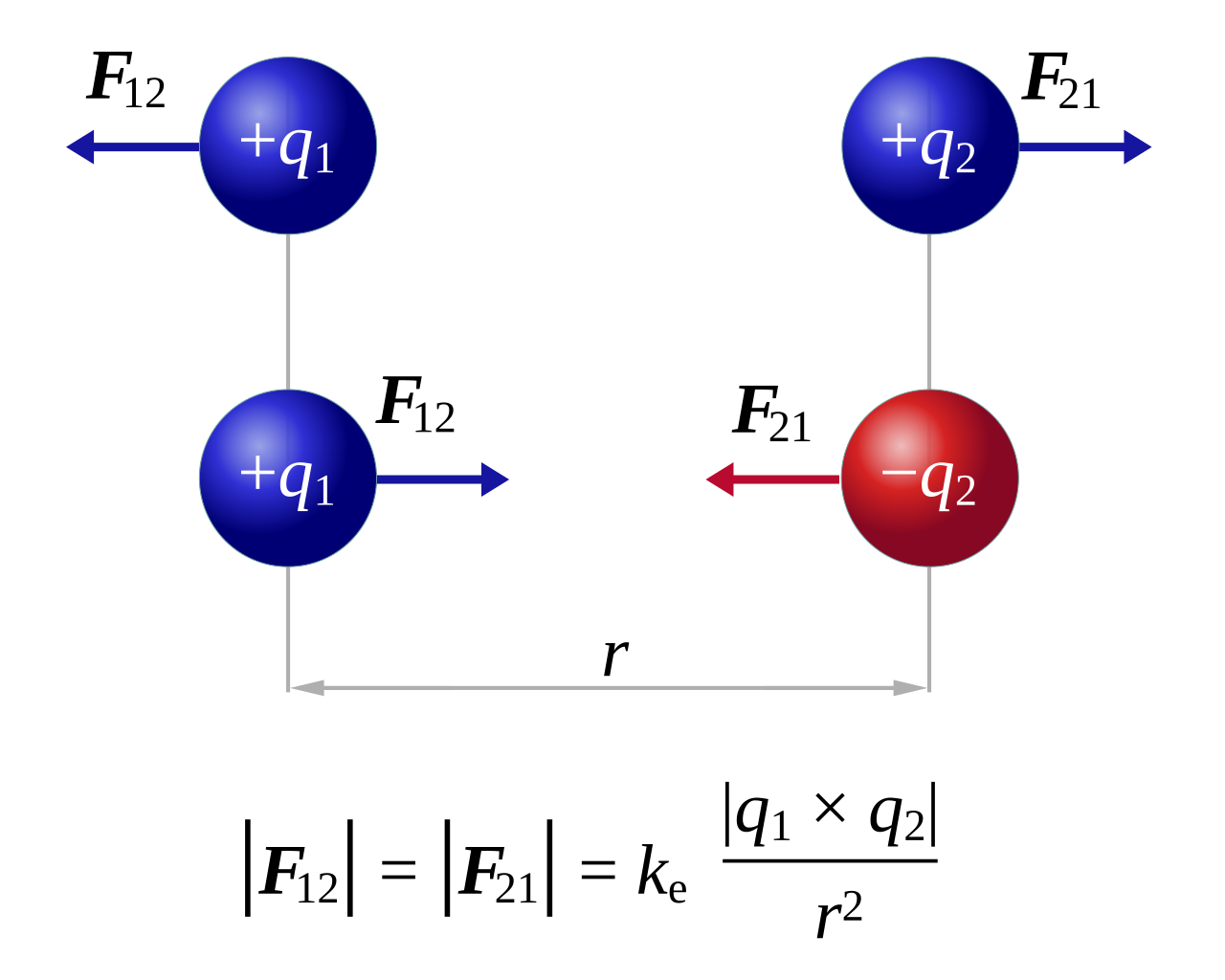
\[k_{e}\] is Coulomb's constant, \[q_{1}\] and \[q_{2}\] are the quantities of each charge, and the scalar \[r\] is the distance between the charges.
The law states that the magnitude, or absolute value, of the attractive or repulsive electrostatic force between two point charges is directly proportional to the product of the magnitudes of their charges and inversely proportional to the square of the distance between them.
\[k_{e}\] was later defined as \[\frac{1}{4\pi\epsilon_{0}}\] where \[\epsilon_{0}\] is another constant called the permittivity of free space (or vacuum). Physicists defined it that way to avoid having to write \[4\pi\] in the Gauss's law equation.
The vector form of this equation is \[\mathbf{F}_{12}=k_{e}\frac{q_{1}q_{2}}{\left\lVert \mathbf{r}_{1}-\mathbf{r}_{2} \right\rVert^{2}}\hat{\mathbf{r}}_{21}=\frac{1}{4\pi\epsilon_{0}}\frac{q_{1}q_{2}}{\left\lVert \mathbf{r}_{1}-\mathbf{r}_{2} \right\rVert^{2}}\hat{\mathbf{r}}_{21}\], where \[\hat{\mathbf{r}}_{21}=\frac{\mathbf{r}_{1}-\mathbf{r}_{2}}{\left\lVert \mathbf{r}_{1}-\mathbf{r}_{2} \right\rVert}\] (unit vector pointing from particle 2 to 1), thus \[\mathbf{F}_{12}\] refers to force on particle 1 from particle 2.
The reason for an inverted unit vector direction is that if the product \[q_{1}q_{2}\] is positive, the force will in the direction pointing from particle 2 to 1 (which is the direction represented by \[\hat{\mathbf{r}}\]). If the product is negative, the negative sign turns \[\hat{\mathbf{r}}\] into \[-\hat{\mathbf{r}}\], which is the direction pointing from particle 1 to 2.
Multiple source charges
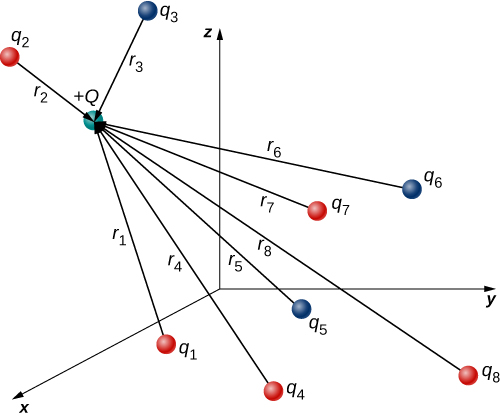
The superposition principle allows Coulomb's law to be extended to include any number of point charges. Given \[N\] charges, the net electric force acting on some other point charge (or referred to as a test charge) is simply the vector sum of each individual electric force exerted on it by each of the individual test charges.
Assume \[\vec{R}\] represents the position of the test charge \[Q\] and \[\vec{R}_{i}\] represents the position of the charge \[i\]. Let \[\vec{r}_{i}=\vec{R}-\vec{R}_{i}\], i.e. the vector pointing from charge \[i\] to the test charge.
Then, the force \[\mathbf{F}\] on a test charge \[Q\] at position \[\vec{R}\] due to a system of \[n\] discrete charges in vacuum is: