physical properties of group 17 elements
Physical properties of group 17 elements
Colour
| Molecule | Colour at room temperature |
|---|---|
| \[\ce{F2}\] | Pale yellow gas |
| \[\ce{Cl2}\] | Yellow-green gas |
| \[\ce{Br2}\] | Dark red liquid |
| \[\ce{I2}\] | Shiny grey-black crystalline solid |
| \[\ce{As2}\] | Radioactive |
Bond strength
As the atoms get bigger, the bonding pair gets further from the nuclei and so it is expected that the strength of the bond to fall.
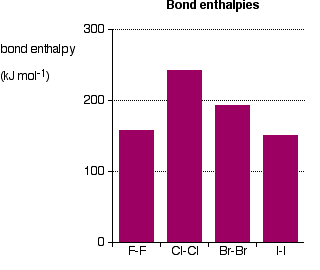
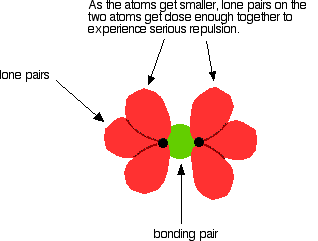
From here, it is shown that \[\ce{Cl-Cl}\], \[\ce{Br-Br}\] and \[\ce{I-I}\] falls in line with the prediction, however \[\ce{F-F}\] falls out of line as the 3 lone pairs in each of the fluorine atoms get close enough together to set up a significant amount of repulsion weakening the bond.
Volatility
Volatility is a material quality which describes how readily a substance vaporizes. At a given temperature and pressure, a substance with high volatility is more likely to exist as a vapour, while a substance with low volatility is more likely to be a liquid or solid.
The volatility of group 17 molecules decrease down the group as the instantaneous dipole-induced dipole interactions between other molecules become stronger due to the increase in electrons. Therefore, an increased amount of energy is required to overcome the intermolecular forces, thus decreasing it's volatility.
Referenced by:
No backlinks found.