metallic bonding
Metallic bonding
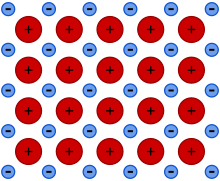
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. Conduction electrons refer to the electrons in a metal that are not bound to individual atoms but are free to move throughout the metal's lattice. These are the electrons responsible for conducting electric current in metals.
Properties
- The freedom of electrons to migrate gives metal atoms, or layers of them, the capacity to slide past each other. Locally, bonds can easily be broken and replaced by new ones after a deformation, which gives rise to metals' characteristic malleability and ductility. This is particularly true for pure elements. In the presence of dissolved impurities, the normally easily formed cleavages may be blocked and the material become harder.
- The atoms in metals have a strong attractive force between them. Much energy is required to overcome it. Therefore, metals often have high boiling points, with tungsten (5828 K) being extremely high.