physical properties of period 3 elements
Physical properties of period 3 elements
Electronic structures
Na: [Ne] 3s1
Mg: [Ne] 3s2
Al: [Ne] 3s2 3p1
Si: [Ne] 3s2 3p2
P: [Ne] 3s2 3p3
S: [Ne] 3s2 3p4
Cl: [Ne] 3s2 3p5
Ar: [Ne] 3s2 3p6
Atomic radius
Unlike a ball, an atom does not have a fixed radius. The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance. The atoms are pulled closely together and so the measured radius is less than if they are just touching. This is what you would get if you had metal atoms in a metallic structure, or atoms covalently bonded to each other. The type of atomic radius being measured here is called the metallic radius or the covalent radius depending on the bonding.
The noble gas at the end of each period should be ignore as argon doesn't form bonds, thus you can only measure their van der Waals radius, a case where the atom is pretty well "unsquashed". All the other atoms are being measured where their atomic radius is being lessened by strong attractions.
From sodium to chlorine, the bonding electrons are all in the \[n=3\] level, screened by the electrons in the first and second levels. The increasing number of protons in the nucleus across the period attracts the bonding electrons more strongly. The amount of screening is constant across Period 3.
Ionic radius
| Ion | Na+ | Mg2+ | Al3+ | Si4+ | P3- | S2- | Cl- |
| Ionic radius (nm) | 0.102 | 0.072 | 0.054 | 0.041 | 0.212 | 0.184 | 0.181 |
The big jump in ionic radius as we get to negative ions is due to an extra layer of electrons. For positive ions, the ions have exactly the same electronic structure, or isoelectronic. However, the number of protons in the nucleus of the ions is increasing. This will exert greater pull towards the electrons causing the ionic radii to fall. For negative ions, they are also isoelectronic, and show similar properties when compared with positive ions, however it is to note that the silicon and chlorine ions have almost the same ionic radius.
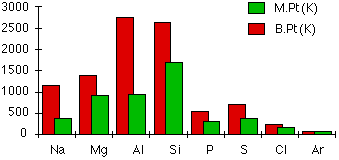
Melting and boiling points
 Temperature in Kelvin.
Temperature in Kelvin.
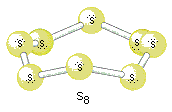
Sodium, magnesium and aluminium are held together by metallic bonds with increasing strength as the number of electrons which each atom can contribute to the delocalised sea of electrons increases, and with the decreasing atomic radius, the metal ions are packed together closer together in the metallic structure, further increasing the bonding strength. Silicon, on the other hand has network covalent structure, which is held together by strong covalent bonds in all three dimensions, requiring a high input of energy. The forces between phosphorus, sulfur and chlorine molecules are van der Waals forces, which is considerably weaker than metallic bonds. Larger molecules have stronger van der Waals forces, therefore the boiling and melting points are \[\ce{S8}>\ce{P4}>\ce{Cl2}>\ce{Ar}\] as shown in the diagram.
\[\ce{S8}\]:
Electrical conductivity
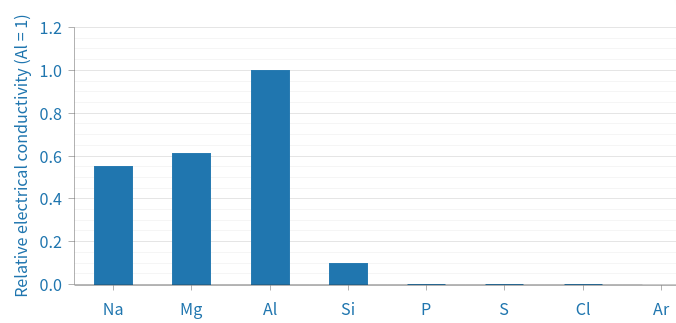
The rising trend can be explained by the increase of delocalised electrons, allowing more electrons to carry charge throughout the system, thus increasing conductivity. Silicon is capable of conducting small amounts of charge, is a semiconductor, so it is not a good conductor or a good insulator.
Referenced by:
No backlinks found.