ion-dipole interactions
Ion-dipole interactions
Ion-dipole and ion-induced dipole forces are similar to dipole-dipole and dipole-induced dipole interactions but involve ions, instead of only polar and non-polar molecules. Ion-dipole and ion-induced dipole forces are stronger than dipole-dipole interactions because the charge of any ion is much greater than the charge of a dipole moment.
An ion-dipole force consists of an ion and a polar molecule interacting. They align so that the positive and negative groups are next to one another, allowing maximum attraction. The polar water molecules surround themselves around ions in water and the energy released during the process is known as hydration enthalpy. The interaction justifies the stability of various ions (like \[\ce{Cu^{2+}}\]) in water.
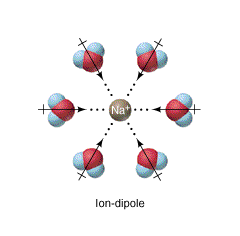
Ion-dipole forces, simply speaking, are the result of the Coulombic electrostatic interactions between an ion and the charged ends of a dipole. Note that here, the term "Intermolecular Force" is a misnomer, even though it is commonly used, as these are the forces between ions with molecules possessing a dipole moment, and ions do not have to be molecular.0
Ion-induced dipole interactions
Similar to dipole-induced dipole interactions, the charge of the ion causes a distortion of the electron cloud in the nonpolar molecule, causing a temporary partial charge. The temporary partially charged dipole and the ion are attracted to each other and form a fleeting interaction.