bond energy
Bond energy
Bond energy (BE) is one measure of the strength of a chemical bond. It is sometimes called the mean bond, bond enthalpy, average bond enthalpy, or bond strength. IUPAC defines bond energy as the average value of the gas-phase bond-dissociation energy (usually at a temperature of 298.15 K or 0 K) for all bonds of the same type within the same chemical species.
Bond energy is the average of all bond-dissociation energies of a single type of bond in a given molecule. The bond-dissociation energies of several different bonds of the same type can vary even within a single molecule.
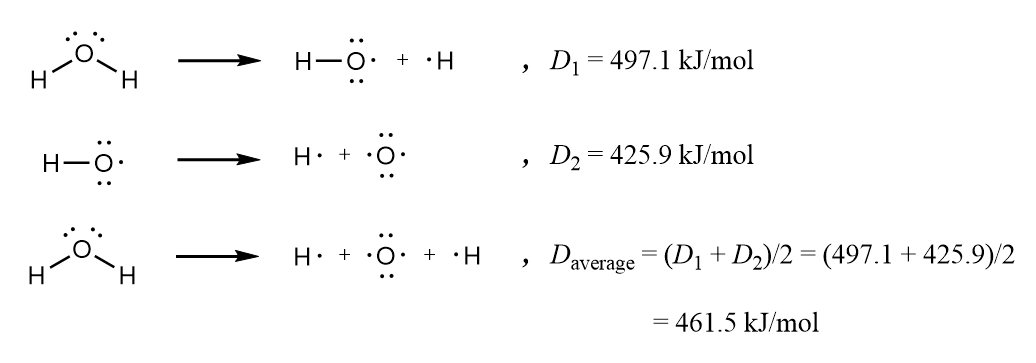
Here, \[D_{1}\] and \[D_{2}\] are the bond-dissociation energies while the bond energy itself is 461.5kJmol-1.
Atomic radii and bond strength
The strength of a bond can be estimated by comparing the atomic radii of the atoms that form the bond to the length of bond itself. For example, the atomic radius of boron is estimated at 85 pm, while the length of the \[\ce{B-B}\] bond in \[\ce{B2Cl4}\] is 175 pm. Dividing the length of this bond by the sum of each boron atom's radius gives a ratio of \[\frac{175}{85+85}=\frac{175}{170}\approx 1.03\]. The ratio is slightly larger than 1, indicating that the bond itself is slightly longer than the expected minimum overlap between the two boron atoms' valence electron clouds. Thus, we can conclude that this bond is a rather weak single bond.
On the other hand, a value lower than 1 indicates that there is a large amount of overlap between the valence electron clouds of the two atoms, which we can conclude that it is a very strong bond. However, this method is only mostly useful to determine covalent bonded compounds.
Ionic compounds
In ionic compounds, the electronegativity of the two atoms bonding together has a major effect on their bond energy. The extent of this effect is described by the compound's lattice energy, where a more negative lattice energy corresponds to a stronger force of attraction between the ions. Generally, greater differences in electronegativity correspond to stronger ionic bonds. For example, the compound sodium chloride (\[\ce{NaCl}\]) has a lattice energy of -786 kJmol-1 with an electronegativity difference of 2.23 between sodium and chlorine. Meanwhile, the compound sodium iodide (\[\ce{NaI}\]) has a lower lattice energy of -704 kJmol-1 with a similarly lower electronegativity difference of 1.73 between sodium and iodine.